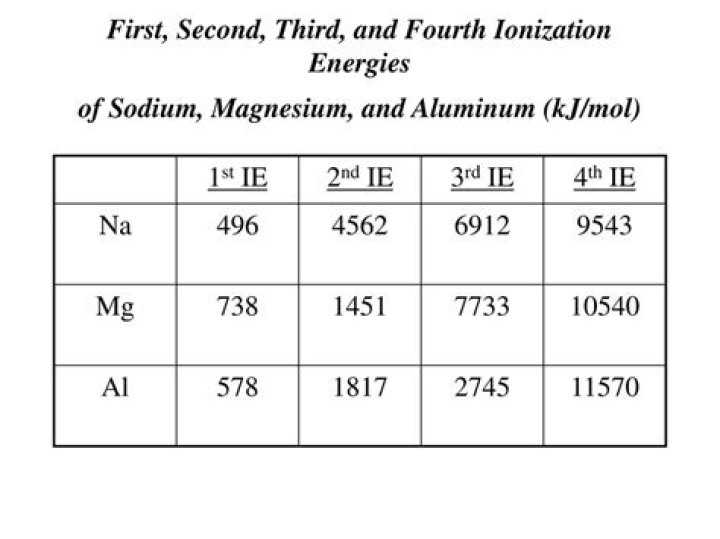
So after it loses one electron it becomes stable and hence more enthalpy is required to remove an electron from the stable atom hence the second ionisation enthalpy of sodium is greater than that of Magnesium.
Why is the second ionization energy of sodium high?
The size of the positive charge increases the higher the number of ionisation is taking place. This means that in the second ionisation energy the outermost electrons will experience more force so the energy required to lose the electron will be higher.
Why is the second ionization energy always greater than the first?
The second ionization energy is always larger than the first ionization energy, because it requires even more energy to remove an electron from a cation than it is from a neutral atom.
Why is the second ionization energy of sodium very high as compared to the first ionization energy of sodium?
since there is a stronger pull from the nucleus, there are less electrons and the shielding effect is smaller; it is harder to pull a second electron from sodium with its full energy level in the second ionization energy.What is the second ionization energy of sodium?
SymbolNaNamesodium2nd45623rd6910.34th9543
What has a higher second ionisation energy neon or sodium?
Hence the outermost electron in the sodium experience a greater nuclear pull and therefore the second ionization enthalpy of sodium is greater than that of first ionization enthalpy of neon.
Why is the second ionization energy greater than the first ionization energy quizlet?
The second ionization energy of Mg is larger than the first because it always takes more energy to remove an electron from a positively charged ion than from a neutral atom. Define “electron shielding.” … Ionisation energy is the energy required to remove an electron from outermost orbital from an isolated gaseous ion.
Why is second ionization energy higher than first ionization energy in alkali metals?
However, after this is done, their nucleus have attained a noble gas stability. And therefore to remove another electron, you would require a very high amount of energy to remove the second electron. This leads to a higher second ionization energy than the first for alkali metals.Why is the ionization energy of sodium less than that of magnesium?
Magnesium atom has a smaller radius and higher nuclear charge than a sodium atom, thus more energy will be required to remove the electron from the same orbital (3s), making the first ionisation energy of magnesium higher than that of sodium.
What is the trend for second ionization energy?Second ionization energy decreases as you go down the group. Third ionization energy decreases as you go down the group. For each element in the Group, the first ionization energy is less than the second ionization energy which is less than the third ionization energy.
Article first time published onWhy ionisation energy of magnesium is higher than Aluminium?
If we compare the ionization energy between magnesium and aluminium, the magnesium has greater ionization energy as it has less no of shells which are closer to the nucleus. Al has one unpaired electron in it’s highest energy orbital (3p), and Mg’s highest energy orbital (3s) the electrons are paired.
Which has the highest second ionization energy?
So, after losing one electron, the configuration is totally octet and is hence a stable structure. So the second ionization energy of sodium will be the highest as that would require extraction of an octet electronic configuration.
Why is the third ionization higher than the second?
The attraction on the outermost electron by the neclues is higher in the case of the third ionization energy since the atom has a positive charge of +2 compared to that at the second ionization energy where the charge on the atom is +1.
What is the difference between first ionization energy and second ionization energy?
First ionization energy: The energy needed to remove the outermost, or highest energy, electron from a neutral atom in the gas phase. Second ionization energy: The energy it takes to remove an electron from a 1+ ion (meaning the atom has already lost one electron and now removing the second).
Which atom would have a second ionization energy very much greater than the first ionization energy?
The second ionization energy of Mg is larger than the first because it always takes more energy to remove an electron from a positively charged ion than from a neutral atom. The third ionization energy of magnesium is enormous, however, because the Mg2+ ion has a filled-shell electron configuration.
Why is the first ionization energy of Na is less than the first ionization energy of CL?
The first ionization energy for sodium is one and one-half times larger than the electron affinity for chlorine. Thus, it takes more energy to remove an electron from a neutral sodium atom than is given off when the electron is picked up by a neutral chlorine atom.
Why does successive ionisation energy increase?
Successive ionization energies increase in magnitude because the number of electrons, which cause repulsion, steadily decrease. … So, the amount of energy needed to remove electrons beyond the valence electrons is significantly greater than the energy of chemical reactions and bonding.
Why does sodium have low ionization energy?
More specifically, elements such as sodium and lithium have low ionization energies because they have only a single electron in their outermost shell. Losing this electron means they are left with a full shell (normally thought of as an octet) which is energetically favorable.
Is second ionization energy?
An element’s second ionization energy is the energy required to remove the outermost, or least bound, electron from a 1+ ion of the element. Because positive charge binds electrons more strongly, the second ionization energy of an element is always higher than the first.
Why does sodium have a higher ionization energy than potassium?
The first ionization energy for sodium is greater than the first ionization energy for potassium due to decrease in the effective nuclear charge.
Why is second ionisation energy of alkali metals higher than alkaline earth metals?
Second ionisation enthalpies of alkali metals are much higher than those of the alkaline earth metals. … Alkali metals have only one valence electron, while alkaline earth metals have two valence electrons. After the removal of one electron from alkali metals, they form M+which is a stable noble gas configuration.
Why the first ionization energy of Be is greater than that of B?
<br> Reason: 2p-orbital is lower in energy than 2s-orbital. Assertion :- The first ionization energy of Be is greater than that of B <br> 2p- orbital is lower in energy when 2s- orbital.
Why is second ionization energy of CR higher than MN?
It can by better understand by considering the electronic configuration of Cr. It form ion by removing the one electron of 4s. … Here we can see there are 5 electrons in 3d which is the more stable state and required highest energy to remove the second electron from Cr. Thus its second ionization energy is higher.
Which element has the highest second ionization energy which element has the highest second ionization energy Na Mg Al Si P?
Out of all the elements given sodium (Na) will have the largest second ionization energy.
Why is the second ionisation energy of magnesium lower than aluminium?
Answer: If we compare the ionization energy between magnesium and aluminium, the magnesium has greater ionization energy as it has less no of shells which are closer to the nucleus. … Therefore, the force of attraction between nucleus and the electrons decreases. Thus, it can remove electrons with less energy.
Why does ionisation energy decrease between magnesium and aluminium?
The first ionisation energy of magnesium is less than that of aluminium. (1) Removal of an electron will disrupt the stable completely-filled 3s sub-shell of magnesium. (2) The 3p electron of aluminium is further from the nucleus compared to the 3s electrons of magnesium.
Does magnesium or aluminum have a higher ionization energy?
So the extra amount of protons means the nucleus holds the outer electrons more strongly so it requires more energy to remove an electron. Aluminium has a lower ionisation energy than Magnesium. This is unexpected as Al has more protons.
Which group has the lowest second ionization energy?
Group 1 elements have the lowest ionization energy due to its effective nuclear charge than the rest of the elements of the period. It further decreases as we move down the group. Hence, we can say that alkali metals have lowest ionization energy.
Which of the following has the highest second ionization energy ie2 )?
Li has the largest second ionization energy (IE2).
Which configuration indicates elements with very high second ionization potential?
Element which has one electron in the valence shell has highest second ionization energy because in this case 2nd electron has to be lost from the inert gas core . Thus option ( c) is correct.
Why is the third ionization higher than the second in magnesium?
In magnesium’s case, however, the third electron would come from the second energy level, more specifically from a 2p-orbital. Since this third electron is located closer to the nucleus for magnesium than for aluminium, you can expect the third ionization energy to be higher in magnesium’s case.