All the amino acids are found in α-helices, but glycine and proline are uncommon, as they destabilize the α-helix. … Because glycine residues have more conformational freedom than other residues, glycine favors the unfolded conformation over the helix conformation. Proline, on the other hand, is too rigid.
What would destabilize an alpha helix?
An α-helix is a right-handed coil of amino-acid residues on a polypeptide chain, typically ranging between 4 and 40 residues. … Amino acids whose R-groups are too large (tryptophan, tyrosine) or too small (glycine) destabilize α-helices.
Why proline and glycine would not occur frequently in the α helical region?
The amino acid side chains face outward, away from the helix axis. The side chains can stabilize or destabilize the helix but are not essential for helix formation. Proline is too rigid to fit into the α-helix, and glycine is too flexible.
Does glycine encourage alpha helix formation?
Surface Exposure of Glycines The forgoing results show that in native-like structures conserved glycine residues are primarily involved in enhancing helix-helix interactions and are not likely to be exposed to the lipid environment.How does glycine affect protein structure?
Conclusions. Glycine and proline residues have a major influence on the kinetics of loop formation in proteins. Glycine accelerates loop formation by decreasing the activation energy, whereas trans prolyl bonds slow loop formation by increasing the barrier height.
What is unique about glycine compared with other naturally occurring amino acids?
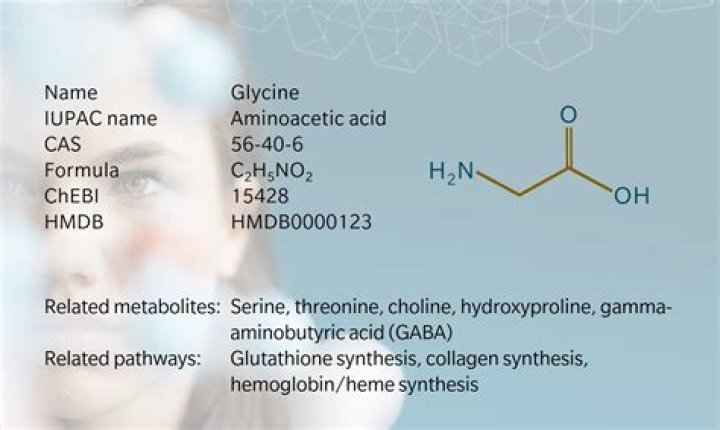
Role in structure: Glycine is a very unique amino acid in that in contains a hydrogen as its side chain (rather than a carbon as is the case in all other amino acids). … What this means is that glycine can reside in parts of protein structures that are forbidden to all other amino acids (e.g. tight turns in structures).
What is L glycine used for?
Glycine is an amino acid with many impressive health benefits. Your body needs glycine to make important compounds, such as glutathione, creatine and collagen. This amino acid may also protect your liver from alcohol-induced damage and improve sleep quality and heart health.
What amino acids make up alpha helix?
An alpha helix is tightly packed, and the end result of this twisting formation is that the amino acid chain will form a rod. The amino acids methionine, alanine, leucine, glutamate, and lysine are highly likely to form an alpha helix. The amino acids proline and glycine are unlikely to form an alpha helix.Which amino acids favor alpha helix?
For example, alanine, leucine, lysine and glutamate favor alpha helix; whereas, beta-branched amino acids threonine, valine, and isoleucine favor beta sheet.
Which amino acids stabilize alpha helix?Four aliphatic side chains occur in the standard complement of amino acids: alanine and leucine are helix stabilizing, whereas isoleucine and valine are weakly destabilizing.
Article first time published onWhich amino acid is most likely to disrupt an alpha-helix?
The answer is e. Pro. The alpha-helix is a helical structure where it is stabilized by the hydrogen bonding of the peptide bonds (the N-H of one…
Why are transmembrane proteins alpha helices?
Figure 2-3. The transmembrane domains of integral membrane proteins are predominantly α-helices. This structure causes the amino acid side chains to project radially. … The outwardly directed residues must be predominantly hydrophobic to interact with the fatty acid chains of lipid bilayers.
Why are proline and glycine common in beta turns?
Proline and glycine residues are statistically preferred at several β-turn positions, presumably because their unique side chains contribute favorably to conformational stability in certain β-turn positions.
How does glycine structure affect its function?
Glycine exerts multiple functions in the central nervous system, as an inhibitory neurotransmitter through activation of specific, Cl–permeable, ligand-gated ionotropic receptors and as an obligatory co-agonist with glutamate on the activation of N-methyl-D-aspartate (NMDA) receptors.
Why glycine is a unique amino acid?
Of the 20 common amino acids, glycine is unique in its lack of any side chain carbons, allowing it greater flexibility.
Why are proline and glycine helix breakers?
Proline and glycine are sometimes known as “helix breakers” because they disrupt the regularity of the α helical backbone conformation; however, both have unusual conformational abilities and are commonly found in turns.
What is the difference between glycine and L glycine?
The key difference between glycine and L glycine is that glycine is an amino acid that makes up proteins, whereas L glycine is an isomer of glycine. Glycine is an amino acid. … Among them, L glycine is the stable and most common isomer in organisms since only the L forms of amino acids are used by cells.
Does glycine increase GABA?
Inhibition of GABA breakdown causes a rise in tissue GABA content and an increase in the activity of inhibitory neurons. … About half of the inhibitory synapses in the spinal cord use glycine; most other inhibitory synapses use GABA.
Is glycine excitatory or inhibitory?
Glycine is the main neurotransmitter in inhibitory interneurons of the spinal cord, brainstem, and in some other brain regions involved in the processing of sensorimotor information and locomotor behavior (51).
Why glycine is a highly conserved amino acid residue in the evolution of proteins?
Answer: Glycine is a highly conserved amino acid residue in the evolution of proteins. … Glycine has the smallest side chain of any amino acid. Its size often is critical in allowing polypeptide chains to make tight turns or to approach one another closely.
Why is glycine the most abundant amino acid?
Glycine is one of the proteinogenic amino acids. … Glycine is integral to the formation of alpha-helices in secondary protein structure due to its compact form. For the same reason, it is the most abundant amino acid in collagen triple-helices.
Does glycine increase the flexibility of the peptide backbone?
Glycine is a large, polar amino acid. Glycine increases the flexibility of the peptide backbone. Glycine is similar to tyrosine in terms of structure and chemical properties. Glycine is special because it contains a -SH group important in protein folding.
Which amino acid Cannot be present in an alpha helix structure?
This is because proline cannot form a regular alpha-helix due to steric hindrance arising from its cyclic side chain which also blocks the main chain N atom and chemically prevents it forming a hydrogen bond.
How many residues separate amino acids that are stabilized by hydrogen bonds in α helices?
An α-helix has 3.6 residues per turn, meaning amino acid side chains that are three or four residues apart are bought together in space and so α-helices are stabilized by hydrogen bond formation between the carbonyl oxygen of one amino acid, and the amide proton of another amino acid four residues further along the …
Why is alpha helix more stable?
α-helix is right handed and is more stable due to inter molecular H bonding between first and fourth amino acid.
Which peptide would be most likely to form an alpha helix?
Peptide c is most likely to form an alpha helix with its three charged residues (Lys, Glu, and Arg) aligned on one face of the helix.
Which type of interactions are responsible for making the alpha helix structure stable?
Hydrogen bonds formed between -NH- group of amino acids in one turn with the >C=O. groups of amino acids belonging to adjacent turn is responsible for making the α−helix structure stable.
How weak interactions stabilizes the alpha helix in proteins?
The alpha helix is stabilized by hydrogen bonds (shown as dashed lines) from the carbonyl oxygen of one amino acid to the amino group of a second amino acid. Because the amino acids connected by each hydrogen bond are four apart in the primary sequence, these main chain hydrogen bonds are called “n to n+4”.
Which amino acid is essentially incompatible with alpha helix and beta sheet formation?
The primary amine on the α carbon of glutamate semialdehyde forms a Schiff base with the aldehyde which is then reduced, yielding proline. When proline is in a peptide bond, it does not have a hydrogen on the α amino group, so it cannot donate a hydrogen bond to stabilize an α helix or a β sheet.
How does pH affect alpha helix?
The secondary structure strongly depends on pH. Thus, at pH above pI (6.8), all the protein structure is in alpha helix. … The sensitivity towards thermal denaturalization is also affected by pH rises.
Which two amino acids are the least likely to occur in the middle of an alpha helical protein structure?
Which of the following amino acids is least likely to be found in the middle of an alpha helix? Explanation: Proline is bound to two alkyl groups thus giving it a planar configuration, giving the nitrogen only the ability to accept hydrogen bonds not donate them.