The known impurities of ASP are already reported, which includes salicylic acid, salsalate (salicylsalicylic acid), acetylsalicylsalicylic acid, 4-hydroxybenzoic acid, 4-hydroxyisophthalic acid and acetylaalicylic anhydride (3, 4).
What are the impurities present in aspirin?
It is often contaminated with salicylic acid, acetic acid, and/or sulfuric acid. Students should not taste their aspirin.
How can you detect impurities in aspirin?
Because today’s aspirin is mass produced in tablet form, Spectrophotometers are a reliable and economical way to keep the purity of Aspirin consistent throughout the manufacturing process. The purity and amount of acetylsalicylic acid in aspirin can be measured using a Visual Spectrophotometer.
What is the most likely impurity in the sample of aspirin?
The main impurity in our crystallized aspirin will be salicylic acid. Salicylic acid will co-precipitate with the aspirin if the procedure is done…What is aspirin composed of?
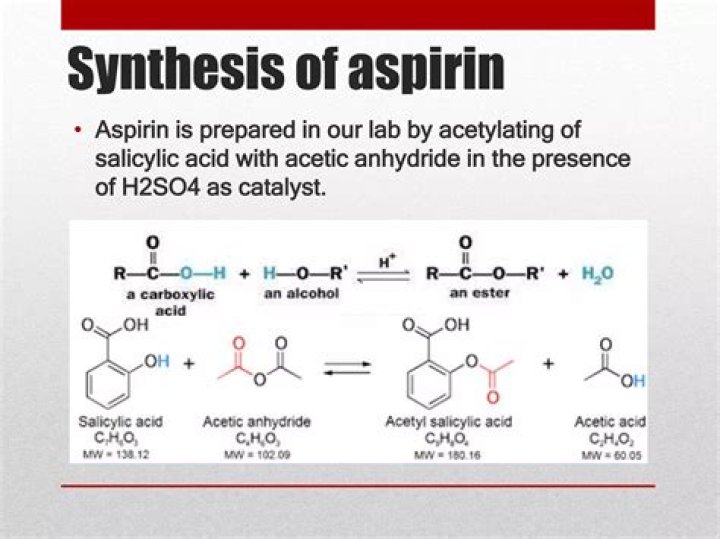
Aspirin is prepared by chemical synthesis from salicylic acid, through acetylation with acetic anhydride. The molecular weight of aspirin is 180.16g/mol. It is odourless, colourless to white crystals or crystalline powder.
What types of impurities could be present in a synthesis?
The three different types of impurities in pharmaceuticals include organic, inorganic, and residual solvents. Most of these impurities occur due to manufacturing processes, degradation, storage conditions, excipients, or contamination.
Is aspirin a polar?
Aspirin is a polar molecule with dipole-dipole attraction bonds and an -OH (hydroxyl) segment as part of a carboxylic acid group.
How does titration determine purity of aspirin?
1 mole of aspirin will react with 2 moles of NaOH So the number of moles of aspirin must be 0.016/2 = 0.008 moles Molecular mass of aspirin (C9H8O4) = 180, so mass of acetylsalicylic acid = 0.008 * 180 = 1.44 g So % purity of aspirin = (1.44/1.504) * 100 = 95.74% The other titration results was calculated the same way.What depth of sample must be loaded into the MP capillary?
Q: How much sample should I put in the capillary tube? A: As a guide, around 2 to 4mm in height. For efficient heat transfer, ensure the sample is well packed in the bottom of the tube by tapping on a hard surface. Prepare all samples to the same height for reproducibility.
What is the percentage purity of aspirin?The aspirin was 99.6 % pure.
Article first time published onWhich is used to detect the presence of impurities in milk?
Soap is added to milk to increase the foaming of milk and thus to have thick milk. Addition of such chemicals will cause health problem especially related to stomach and kidneys. Soap can be detected by adding phenolphthalein indicator to the adulterated milk.
What are the physical and chemical properties of aspirin?
Aspirin, an acetyl derivative of salicylic acid, is a white, crystalline, weakly acidic substance, with a melting point of 136 °C (277 °F), and a boiling point of 140 °C (284 °F).
What is the pKa of aspirin?
These include aspirin (pKa 3.5), the anticancer compound methotrexate (pKa 3.8, 4.8 and 5.6) and the diuretic (previously called frusemide in the UK; pKa 3.9).
Is aspirin an organic or inorganic compound?
The chemical name for aspirin is acetylsalicylic acid. Its chemical formula is C-9, H-8, O-4. Acetylsalicylic acid is an example of an organic compound, which means it contains carbon.
Is aspirin soluble?
Aspirin is only slightly soluble in water so if you add it to the reaction mixture the product would rather clump together than be in the water phase. This causes the solid to begin to form or ‘precipitate’.
What is the solubility of aspirin?
It has a limited solubility in water, which amounts to 2–4 mg/mL and its solubility varies significantly with temperature [1]. Aspirin is more soluble in ethanol, ethyl ether, chloroform, sodium hydroxide solution, and sodium carbonate solution than in water.
Why is aspirin so polar?
Aspirin has an aromatic ester and an aromatic carboxylic acid functional group; therefore, it has the potential of being very polar. Since the two functional groups are ortho to each other, however, hydrogen bonding forces on the acidic proton cancel out with forces on the ester, and aspirin elutes first.
What are pharmaceutical impurities?
Pharmaceutical impurities are the unwanted chemicals that remain with active. pharmaceutical ingredients (APIs) or drug product formulations. The impurities. observed in drug substances may arise during synthesis, or may be derived from.
What kind of impurities are present in solid substance?
Impurities are chemical substances inside a confined amount of liquid, gas, or solid, which differ from the chemical composition of the material or compound. Impurities are either naturally occurring or added during synthesis of a chemical or commercial product.
What are the sources of impurities in pharmaceutical?
The various sources of impurity in pharmaceutical products are — reagents, heavy metals, ligands, catalysts, other materials like filter aids, charcoal, and the like, degraded end products obtained during \ after manufacturing of bulk drugs from hydrolysis, photolytic cleavage, oxidative degradation, decarboxylation, …
What does melting point tell you about purity?
Melting point (m.p.) analysis can also provide information about the purity of a sample. A substance (solid) containing soluble impurities usually melts at a lower temperature than the pure compound. … In general, the smaller the range of melting temperatures, the higher the purity of the sample.
Why is it incorrect to speak of a melting point?
In pure crystalline organic substances the range is nearly 0.5 deg C. Hence actually speaking the melting point is a range over which the melting process is taking place and is not a fixed temperature . As the amount of impurities in the substance increases ,it also increases the range of melting point.
What is the effect of impurities in the sample?
The presence of impurities in a substance results in a lower melting point due to a process called melting point depression. Melting point depression is the reason why adding salt to frozen streets helps to melt the ice. Melting point depression occurs due to the nature of a material’s solid state.
Why is my aspirin not pure?
The melting point range for the acetylsalicylic acid obtained is 144°C-172°C, it is because it contains impurities. The theoretical yield of aspirin is 2.628g, but the actual yield of aspirin obtained is 0.8953g. therefore, the percentage yield of aspirin is 34.07%.
How do you find the mass of pure aspirin?
For the sample data, the mass of synthesized aspirin is 1.96 g. Corrected for purity, is: (1.96 × 0.75) = 1.47 g. The percent yield is: (1.47 g ÷ 2.63 g) × 100 = 55.9%.
How much acetylsalicylic acid is in an aspirin tablet?
Each tablet contains 75 mg acetylsalicylic acid.
How does titration determine purity?
Conversely, the percent purity of an impure sample of a chemical of unknown percent purity can be determined by reaction with a pure compound as in an acid-base titration. Percent purity can also be determined, in theory, by measuring the amount of product obtained from a reaction.
Is 60 pure aspirin a good yield?
Reactants may be impure, reactions may not go to completion, or given reactions may have to compete with several smaller side reactions. In fact, in the laboratory, if you get 60% of the expected amount of product, that is considered very good.
Is NaOH solution hygroscopic?
Standard NaOH Solution Solid sodium hydroxide is hygroscopic, which means that it absorbs moisture from the atmosphere.
How do you test for impurities in milk?
- Put a drop of milk on a polished slanting surface.
- Pure milk either stays or flows slowly leaving a white trail behind.
- Milk adulterated with water will flow immediately without leaving a mark.
What is adulteration of honey?
Direct adulteration of honey is commonly performed by the direct addition of a certain amount of sucrose syrup into the honey. The source of sucrose syrup could be sugar beet, HFCS, maltose syrup, or industrial sugar syrups (glucose and fructose) obtained from heat, enzyme, or acid treatment of starch [74].