Trigonal planar molecular geometryPoint groupD3hCoordination number3Bond angle(s)120°μ (Polarity)0
What molecules are trigonal pyramidal?
Trigonal pyramidal is a molecular shape that results when there are three bonds and one lone pair on the central atom in the molecule. Molecules with an tetrahedral electron pair geometries have sp3 hybridization at the central atom. Ammonia (NH3) is a trigonal pyramidal molecule.
Is nh3 a trigonal planar molecule?
It is trigonal pyramidal because of the lone pair of electrons associated with the central nitrogen atom.
Is BH3 a trigonal planar?
An example of trigonal planar electron pair geometry (E. P. G.) and molecular geometry is BH3. … This is trigonal planar geometry. The molecule all in a plane and is two dimensional.Is trigonal planar linear?
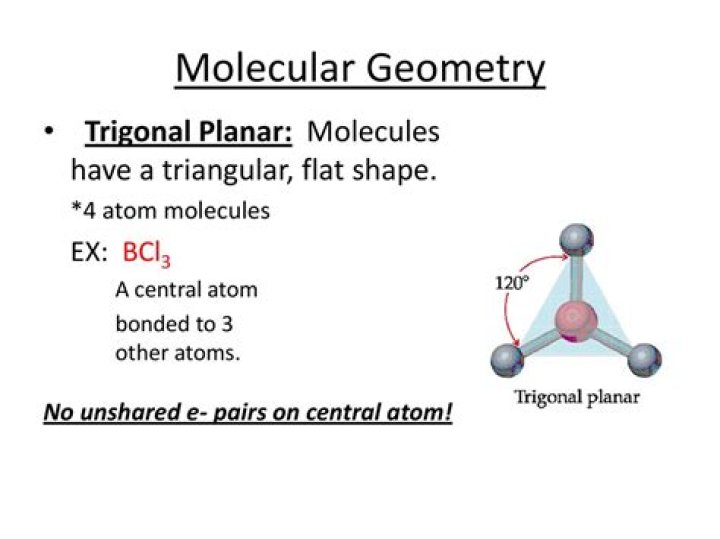
Linear: a simple triatomic molecule of the type AX2; its two bonding orbitals are 180° apart. Trigonal planar: triangular and in one plane, with bond angles of 120°. … Trigonal bipyramidal: five atoms around the central atom; three in a plane with bond angles of 120° and two on opposite ends of the molecule.
Is NO3 trigonal planar?
In essence, nitrate has 3 electron domains with zero lone pairs. Thus, NO3- molecular geometry is trigonal planar and is slightly bent. The bond angle is 120°.
What does a trigonal planar look like?
A trigonal planar compound has a central atom attached to three atoms arranged in a triangular shape around the central atom. All four atoms lie flat on a plane. … We can see that the bond angles between the atoms attached to the central atom are all 120 degrees.
Is CH4 trigonal planar?
$\text{C}{{\text{H}}_{4}}$Central atomCarbonValence electron on central atom4Is CH3 trigonal planar?
CH3 (methyl free radical) has a planar structure with sp2 hybridization of ‘C’ atom. … (methyl carbonium ion) also has trigonal planar structure (sp2). (methyl carbanion) has tetrahedral structure (sp3) and one of the hybrid orbitals contains the lone pair of electrons.
Is H2CO trigonal planar?The central atom of H2CO, has three regions of electron density around it. The regions of electrons making a trigonal planar shape, giving a bond angle of 120°. All three of these regions of electrons are bonding so the arrangement of the bonds/molecular shape is trigonal planar.
Article first time published onIs bcl3 trigonal planar?
BCl3 Molecular Geometry And Bond Angles If we look at the structure, BCl3 molecular geometry is trigonal planar. The bond angle is 120o. The central atom also has a symmetric charge around it and the molecule is non-polar.
Is trigonal planar the same as trigonal pyramidal?
What is the difference between Trigonal Planar and Trigonal Pyramidal? In trigonal planar, there are no lone pair electrons in the central atom. But in trigonal pyramidal there is one lone pair at the central atom. … In trigonal planar, all the atoms are in one plane but, in trigonal pyramidal they are not in one plane.
What angle is trigonal planar?
Number of Electron GroupsElectron-Group GeometryIdeal Bond Angles2linear180°3trigonal-planar120°120°4tetrahedral109.5°
How do you find planar geometry?
The basic way to decide is to look at the hybridisation of compounds. If the hybridisation is sp2 then the compound is planar. The shape of compound is determined by it’s hybridisation. If it is sp then it’s linear, sp2 has a shape of triangular planar, sp3 has a shape of tetrahedral,sp3d as trigonal bi pyramid,etc.
Is no2 trigonal planar?
The nitrogen and and one oxygen are bonded through a double bond which counts as “one electron pair”. Hence the molecule has three electron pairs and is trigonal planar for electron pair geometry. … Nitrogen dioxide is a main component of photochemical smog air pollution.
Is trigonal planar 2d or 3d?
The central and surrounding atoms in a trigonal planar molecule lie on one plane (hence the term planar). This gives it more of a two-dimensional shape than three-dimensional.
Which of the following molecule has planar geometry?
O2SF2.
Is SO2 a shape?
SO2 Lewis Structure- Key Points The electron geometry of SO2 is formed in the shape of a trigonal planner. The three pairs of bonding electrons arranged in the plane at an angle of 120-degree.
Is co32 planar?
3 that the molecular geometry of CO 3 2− is trigonal planar with bond angles of 120°.
Is no3 a molecular shape?
Name of the MoleculeNitrateGeometryTrigonal Planar
Is AlCl3 trigonal planar?
The molecular geometry of AlCl3 is trigonal planar with each Al-Cl bond angeled 120° to each other and its electron geometry is also trigonal planar.
Does CH3+ have trigonal planar shape?
So, the correct answer is “Option A”.
Is CF3 radical planar?
Of the two free radicals, methyl (•CH3) and trifluoromethyl (•CF3), one is planar, the other pyramidal. … In this extreme, the orbital is 25% s in character; therefore, it is the CF3• which is pyramidal and the CH3• which is planar.
Is PH3 trigonal planar or pyramidal?
Its molecular shape is pyramidal as P has 5 valence e- of which 3 shared with H and 2 remain as 1 lone pair so, hybridization comes out to be sp3(3+1). The shape is called trigonal pyramidal.
What shape is PH3?
The shape of phosphine is pyramidal ; i.e. it is an analogue of ammonia.
Does PH3 have tetrahedral geometry?
Shape of PH3 is trigonal pyramidal. Molecular geometry around phosphorous atom is tetrahedral. Total valence electrons pairs around phosphorous atom is four.
Is CH4 trigonal pyramid?
For example; four electron pairs are distributed in a tetrahedral shape. If these are all bond pairs the molecular geometry is tetrahedral (e.g. CH4). If there is one lone pair of electrons and three bond pairs the resulting molecular geometry is trigonal pyramidal (e.g. NH3).
What is the shape of ch3?
It is trigonal pyramidal and sp3 hybridized.
What is geometry of CH4 molecule?
The molecular geometry of CH4 is tetrahedral as the carbon central atom has no lone pair and is attached to the four hydrogen atoms with the help of a single covalent bond.
Why is CH2O trigonal planar?
The electron geometry of CH2O is also trigonal planar because there is no lone pair present on the central atom(carbon), hence, only bonded pair will be counted for determining the shape of CH2O.
Is NCl3 trigonal planar?
The molecular geometry of NCl3 is trigonal pyramidal and its electron geometry is tetrahedral. NCl3 lewis dot structure contains 1 lone pair and 3 bonded pairs. The net dipole moment of Nitrogen trichloride is 0.6 D.