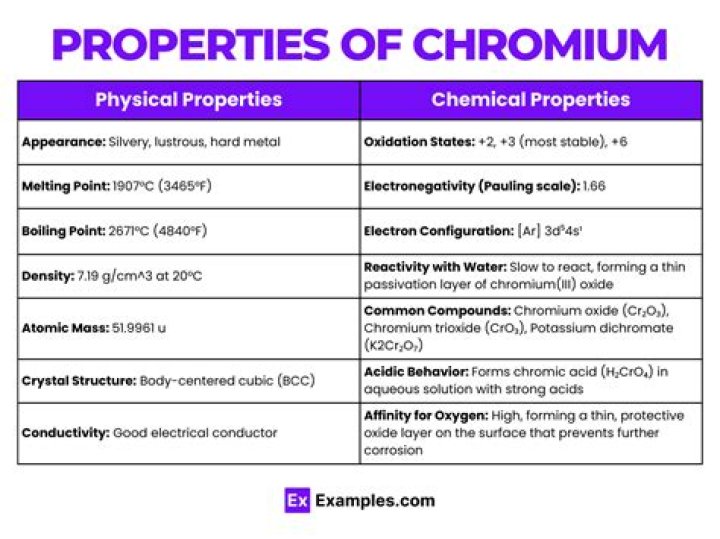
Chromium is a lustrous, brittle, hard metal. Its colour is silver-gray and it can be highly polished. It does not tarnish in air, when heated it borns and forms the green chromic oxide. Chromium is unstable in oxygen, it immediately produces a thin oxide layer that is impermeable to oxygen and protects the metal below.
What are 2 physical properties of chromium?
Chromium Properties: Chromium has a melting point of 1857+/-20°C, a boiling point of 2672°C, a specific gravity of 7.18 to 7.20 (20°C), with valences usually 2, 3, or 6. The metal is a lustrous steel-gray color which takes a high polish. It is hard and resistant to corrosion.
What are the properties and uses of chromium?
Uses and properties A hard, silvery metal with a blue tinge. Chromium is used to harden steel, to manufacture stainless steel (named as it won’t rust) and to produce several alloys. Chromium plating can be used to give a polished mirror finish to steel.
Is chromium a physical or chemical property?
Chromium is a chemical element with Cr as its symbol. It belongs to group 6, periodic number 4 of the periodic table. Its atomic number is 24. Chromium is a steely-gray lustrous, brittle, hard metal.Is chromium brittle or malleable?
Chromium is a hard, gray metal that is valued for its incredible resistance to corrosion. Pure chromium is magnetic and brittle, but when alloyed can be made malleable and polished to a bright, silvery finish.
What are the properties of Chrome?
atomic number24melting point1,890 °C (3,434 °F)boiling point2,482 °C (4,500 °F)specific gravity7.20 (28 °C)oxidation states+2, +3, +6
What are the properties of chromium Class 8?
- 1907 °C. Period.
- 2672 °C. Block.
- 7.19 g.cm-3 at 20°C. Atomic number.
- 52Cr. Electron configuration.
How do you identify chromium?
Chromium is a chemical element with the symbol Cr and atomic number 24. It is the first element in group 6. It is a steely-grey, lustrous, hard, and brittle transition metal.What are titanium's properties?
Titanium is a transition metal light with a white-silvery-metallic colour. It is strong, lustrous, corrosion-resistant. Pure titanium is not soluble in water but is soluble in concentrated acids.
Is chromium soluble or insoluble?The solubility of chromium compounds varies, depending primarily on the oxidation state. Trivalent chromium compounds, with the exception of acetate, hexahydrate of chloride, and nitrate salts, are generally insoluble in water (Table 4-2).
Article first time published onWhat are the properties of chromium which make it useful for electroplating?
Chromium has a good shine, it resists scratches and is corrosion resistant.
What are the physical properties of sodium?
- Symbol: Na.
- Melting point: 97.794°C.
- Boiling point: 882.940°C.
- Density (g cm−3): 0.97.
- Atomic mass: 22.990.
- Atomic number: 11.
- Electronegativity (Pauling Scale): 0.93.
- Classification: Alkali metal, Group I metal.
Which properties of chromium metal make it suitable for electroplating?
Chromium is highly resistant to corrosion and rusting which helps to increase the durability of the metal objects also chromium prevents scratches and has a shiny silver-like appearance. Due to these factors chromium metal is generally electroplated on car parts such as bumpers and bicycle handlebars made of steel.
Why is chromium malleable?
Most metals are malleable because the atoms can roll over each other and retain the structure of the crystal.
Is chromium a conductor?
Chromium. Not a very good electrical conductor by any means, but it does have its uses in this field. Chromium Boride (CrB) is used as a high temperature electrical conductor. There are many other uses for this metal.
Is chromium a refractory metal?
A narrowly defined class of refractory metals would include metals with melting points > 2000°C: niobium, chromium, molybdenum, tantalum, tungsten, and rhenium [1], while a wider class would also include those with melting points above 1850°C: vanadium, hafnium, titanium, zirconium, ruthenium, osmium, rhodium, and …
What is the Amu of chromium?
Atomic NumberSymbolAtomic Weight (amu, g/mol)24Cr51.99625Mn54.938026Fe55.84728Ni58.70
What are coppers uses?
Most copper is used in electrical equipment such as wiring and motors. This is because it conducts both heat and electricity very well, and can be drawn into wires. It also has uses in construction (for example roofing and plumbing), and industrial machinery (such as heat exchangers).
Which of the following is not the property of chromium film?
7. Which of the following is not the property of Chromium film? Explanation: The Chromium film is non-porous, insoluble, self-healing, and continuous.
What type of solid is chromium?
Atomic Mass51.996uYear Discovered1797
What is the difference between Chrome and Chromium?
Chromium is an open-source and free web browser that is managed by the Chromium Project. In comparison, Google Chrome is a proprietary browser developed and managed by Google. Unlike Chromium, Google Chrome offers built-in support for media codecs like MP3, H. 264, and AAC, as well as Adobe Flash.
Is chromium a steel?
Chromium is a powerful alloying element in steel. It strongly increases the hardenability of steel, and markedly improves the corrosion resistance of alloys in oxidizing media. … Stainless steels may contain in excess of 12% chromium.
What are 5 physical properties of titanium?
- Element Classification: Transition Metal.
- Density (g/cc): 4.54.
- Melting Point (K): 1933.
- Boiling Point (K): 3560.
- Appearance: Shiny, dark-gray metal.
- Atomic Radius (pm): 147.
- Atomic Volume (cc/mol): 10.6.
- Covalent Radius (pm): 132.
What are properties of tungsten?
Tungsten metal has a nickel-white to grayish lustre. Among metals it has the highest melting point, at 3,410 °C (6,170 °F), the highest tensile strength at temperatures of more than 1,650 °C (3,002 °F), and the lowest coefficient of linear thermal expansion (4.43 × 10−6 per °C at 20 °C [68 °F]).
What are three characteristics of titanium?
Titanium is well known for its properties of lightness, strength, and high-corrosion-resistance, but it is not so well known that there are other propertiess and what they are really capable of.
Is chromium a mineral or vitamin?
Chromium is a mineral our bodies use in small amounts for normal body functions, such as digesting food. Chromium exists in many natural foods including brewer’s yeast, meats, potatoes (especially the skins), cheeses, molasses, spices, whole-grain breads and cereals, and fresh fruits and vegetables.
How does chromium enter the body?
When you breathe air containing chromium, some of the chromium will enter your body through your lungs. Some forms of chromium can remain in the lungs for several years or longer. A small percentage of ingested chromium will enter the body through the digestive tract.
How is chromium extracted?
Chromium is generally extracted from its ores by oxidation in the form of chromate, in which state its valence is six. The product of this reaction is then treated to extract the chromate. … In the present state of the art, the commonly used process for oxidation of chromium ores is known as oxidizing alkaline roasting.
Why is chromium in water?
Chromium-6 occurs naturally in the environment from the erosion of natural chromium deposits. It can also be produced by industrial processes. There are demonstrated instances of chromium being released to the environment by leakage, poor storage, or inadequate industrial waste disposal practices.
How does chromium get in water?
In addition to natural sources, hexavalent chromium enters drinking water sources through discharges of dye and paint pigments, wood preservatives, chrome plating wastes, and leaching from hazardous waste sites.
How does chromium react in water?
Elementary chromium does not react with water at room temperature. Many chromium compounds are relatively water insoluble. Chromium (III) compounds are water insoluble because these are largely bound to floating particles in water. Chromium (III) oxide and chromium (III) hydroxide are the only water soluble compounds.