Atomic number12Electronegativity according to Pauling1.2Density1.74 g.cm -3 at 20 °CMelting point650 °CBoiling point1107 °C
Which is a physical property of magnesium?
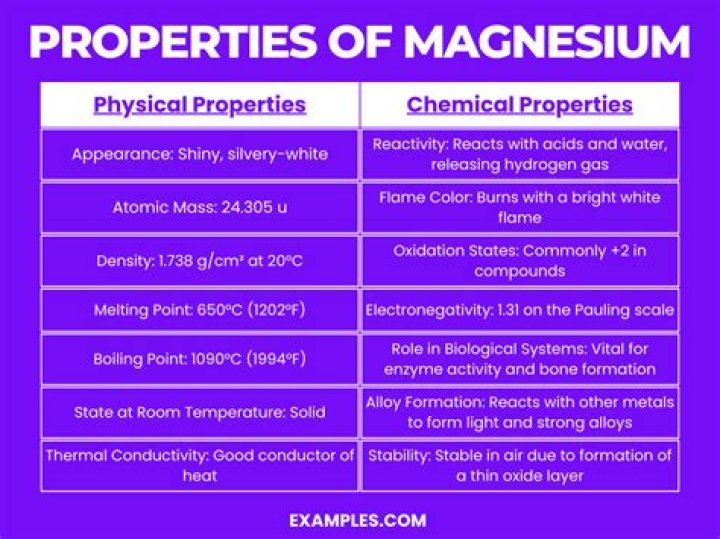
Physical Properties: Magnesium is a shiny, silver or gray colored metal that is light in weight and strong. Th density of magnesium is 1.738 g/mL, which means the metal will sink in water, but it is still relatively light weight.
What are the physical and chemical properties of magnesium oxide?
Compound FormulaMgOAppearanceWhite PowderMelting Point2,852° C (5,166° F)Boiling Point3,600° C (6,512° F)Density3.58 g/cm3
What are the chemical properties for magnesium?
atomic number12melting point650 °C (1,202 °F)boiling point1,090 °C (1,994 °F)specific gravity1.74 at 20° C (68 °F)oxidation state+2What are 3 physical properties of magnesium ribbon?
Molecular Weight24.31AppearanceGrayMelting Point650 °CBoiling Point1090 °CDensity1738 kg/m3
What are 4 physical properties of magnesium?
Atomic number12Electronegativity according to Pauling1.2Density1.74 g.cm -3 at 20 °CMelting point650 °CBoiling point1107 °C
What are the 2 properties of magnesium bromide?
Compound FormulaBr2MgAppearanceWhite light brown solidMelting Point711° C (1,312° F)Boiling PointN/ADensity3.72 g/cm3
Which is chemical property?
A chemical property is a characteristic of a particular substance that can be observed in a chemical reaction. Some major chemical properties include flammability, toxicity, heat of combustion, pH value, rate of radioactive decay, and chemical stability.What is the physical properties of magnesium hydroxide?
NamesAppearanceWhite solidOdorOdorlessDensity2.3446 g/cm3Melting point350 °C (662 °F; 623 K) decomposes
What are 5 physical properties of magnesium oxide?Magnesium can be commercially produced from carnallite, brucite, magnesite, olivine and talc. Magnesium oxide nanoparticles are odorless and non-toxic. They possess high hardness, high purity and a high melting point. Magnesium oxide nanoparticles appear in a white powder form.
Article first time published onWhat are the chemical properties of magnesium hydroxide?
Magnesium hydroxide has the chemical formula Mg(OH)2 and the molecular weight of 58.3197 g/mol. Its CAS number is 1309-42-8 and its density is 2.345 g/cm3. It decomposes when heated to 360 °C and forms the oxide, MgO. It is very slightly soluble in water at 0.00122 g/100 ml and has a solubility product of 5.61 × 10−12.
Does magnesium and magnesium oxide have the same chemical properties?
Magnesium is an element with a specific atomic number (12) and symbol (Mg). … On the other hand, magnesium oxide is a compound derived when magnesium combines with oxygen. It has its own chemical formula (MgO) which is formed due to an ionic bond. However, this bond can be broken by chemical decomposition.
What are 3 interesting facts about magnesium?
- Magnesium is the metal ion found at the center of every chlorophyll molecule. …
- Magnesium ions taste sour. …
- Adding water to a magnesium fire produces hydrogen gas, which can cause the fire to burn more fiercely.
- Magnesium is a silvery-white alkaline earth metal.
What is the chemical equation for burning magnesium ribbon?
The equation is: Magnesium + oxygen → magnesium oxide. 2Mg + O2 → 2MgO.
Is magnesium a metal?
A silvery-white metal that ignites easily in air and burns with a bright light. Magnesium is one-third less dense than aluminium.
What is magnesium do?
Magnesium is a nutrient that the body needs to stay healthy. Magnesium is important for many processes in the body, including regulating muscle and nerve function, blood sugar levels, and blood pressure and making protein, bone, and DNA.
What color is magnesium?
Magnesium is one of the alkaline-earth metals, and is one of the most common elements in the Earth’s crust. In its pure form, it is silvery white, and relatively soft. It burns in air with a brilliant white light, and for this reason is often used in flares and fireworks.
What is the chemical name of magnesium bromide?
PubChem CID522691Molecular FormulaBr2MgSynonymsMagnesium bromide (MgBr2) magnesium;dibromide MgBr2 MFCD00011105 Magnesium bromide, 98% More…Molecular Weight184.11Component CompoundsCID 260 (Hydrogen bromide) CID 5462224 (Magnesium)
What is the chemical symbol of gold?
Gold; it’s chemical symbol is Au and it’s the most malleable and ductile of the known metals. Gold is the highly sought-after because of it’s valuable uses. Gold symbol on the periodic table is Au that was used because of its Latin name aurum.
Is magnesium an insulator or conductor?
HydrogenN/AConductorMagnesiumConductorSemiconductorAluminumConductorInsulatorSiliconSemiconductorN/APhosphorusConductorConductor
Why magnesium is a metal?
The following are the three reasons that makes magnesium a metal: It is a good conductor of electricity. Magnesium is an electropositive element. The ionization enthalpy of magnesium is low.
What is the meant by the term chemical formula?
Chemical formula are used to describe the types of atoms & their numbers in an element on compound. The atoms of each element are represented by one or two different letters. A set of chemical symbols showing the elements present in a compound & their relative proportions.
What are physical and chemical properties?
Physical properties are those that can be observed without changing the identity of the substance. … Properties that describe how a substance changes into a completely different substance are called chemical properties. Flammability and corrosion/oxidation resistance are examples of chemical properties.
What are four physical properties?
Familiar examples of physical properties include density, color, hardness, melting and boiling points, and electrical conductivity. We can observe some physical properties, such as density and color, without changing the physical state of the matter observed.
What are the examples of physical properties?
- Density.
- color.
- temperature.
- boiling point.
- melting point.
- odor.
- texture.
What is the chemical composition of magnesium precipitate which is crystalline in appearance?
4.2 Struvite precipitation. Struvite is a crystalline solid composed of magnesium, ammonia, and phosphate at equimolar concentrations (MgNH4PO4⋅6H2O).
What type of chemical bond is MgO?
Figure 1 The ionic bonding in magnesium oxide (MgO).
Is magnesium hydroxide ionic or covalent?
Is magnesium hydroxide an ionic or a covalent compound? – Quora. Mg(OH)2 is most typically identified as an ionic compound. That is, the Mg atoms are viewed to be Mg^2+ ions, while there are also two OH^- ions. This formulation satisfies the Lewis octet rule, and classifies the compound as ionic.
What is the nature of magnesium hydroxide Class 7?
Answer: The nature of magnesium hydroxide is as the mineral brucite.
How many atoms are in magnesium hydroxide?
In a formula unit of Mg(OH)2, there are one magnesium ion and two hydroxide ions; or one magnesium, two oxygen, and two hydrogen atoms.
Does magnesium oxide have physical and chemical properties similar to oxygen and magnesium?
Magnesium reacts explosively with oxygen to form magnesium oxide. … Magnesium oxide has physical and chemical properties similar to both oxygen and magnesium. A student reports the melting point of a gas as -295 degrees C.