To Determine Chair Conformation Stability, Add Up The A-Values For Each Axial Substituent. The Lower The Number, The More Stable It is.
Which cyclohexane is more stable?
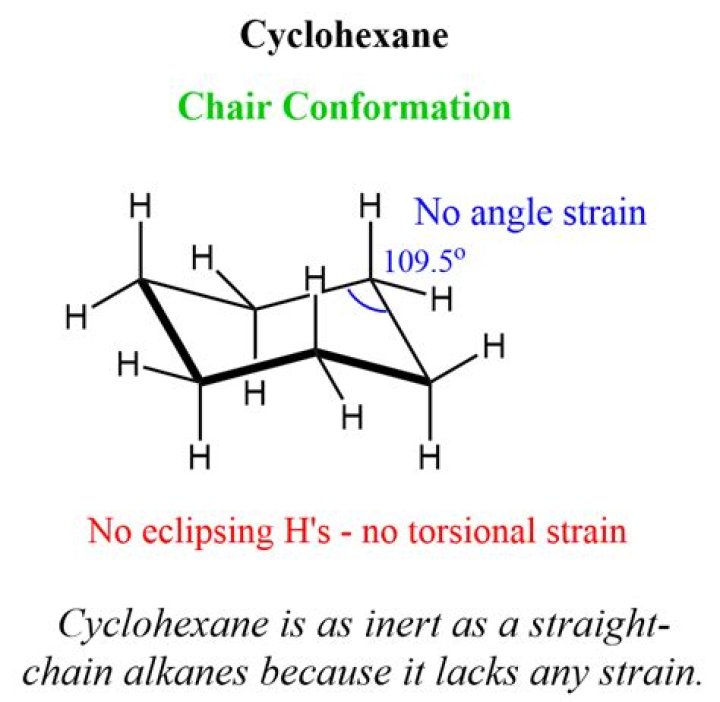
The chair conformation is the most stable conformation of cyclohexane.
How do you know which conformation is more stable?
To find the most stable conformation, we choose the form with the least number of large axial groups; the least stable will have the most number of axial groups.
Which conformation of cyclohexane has highest stability?
The chair conformation is the most stable conformer. At 25 °C, 99.99% of all molecules in a cyclohexane solution adopt this conformation.Which of cyclohexane forms are stable Why?
Which is the most stable conformation of cyclohexane? The chair form shown to the right is the most stable conformation of cyclohexane. The C-C-C bonds are very similar to 109.5o, so they are almost free from angle pressure. It is also a completely staggered conformation, and is therefore free of torsional stress.
How do you know which chair is more stable?
To Determine Chair Conformation Stability, Add Up The A-Values For Each Axial Substituent. The Lower The Number, The More Stable It is.
Which is more stable cyclohexane or hexane?
Since cyclohexane molecules are closely packed, the molecular interactions are much stronger than that of hexane. Therefore, the energy required to break the bonds and evaporate it is much higher. Hence, cyclohexane has a higher boiling point.
Why is chair conformation of cyclohexane more stable?
The chair conformation is more stable because it does not have any steric hindrance or steric repulsion between the hydrogen bonds. By drawing cyclohexane in a chair conformation, we can see how the H’s are positioned. … These are hydrogens in the axial form. These hydrogens are in an equitorial form.Which of the following conformation of cyclohexane is least stable?
The least stable conformation of cyclohexane is half chair conformation.
Which conformation has highest stability?hydrocarbons. …with respect to the other—the eclipsed conformation is the least stable, and the staggered conformation is the most stable. The eclipsed conformation is said to suffer torsional strain because of repulsive forces between electron pairs in the C―H bonds of adjacent carbons.
Article first time published onWhich of the following conformation has highest stability Mcq?
D. The chair form is the most stable conformation of cyclohexane. The C−C−C bonds are very similar to 109.5o , so they are almost free from angular strain.
Which of the following conformer of cyclohexane is the most unstable?
Answer: Half chair form. Explanation: It is the conformer of cyclohexane having the highest enegry in the energy profile diagram of cyclohexane.
Which is more stable cyclohexane or cyclohexene?
Another difference between cyclohexane and cyclohexene is that the cyclohexane is relatively stable, thus, is less reactive while the cyclohexene is relatively unstable, thus, can undergo reactions due to the presence of a double bond in the ring structure.
Why is cyclohexane less reactive than hexane?
Therefore, chemically cyclohexane is more reactive than n-hexane, mainly because of strain in cyclohexane. [1] are similar to alkanes in their general physical properties, but they have higher boiling points, melting points, and densities than alkanes.
How can you distinguish between cyclohexane and hexane?
The key difference between hexane and cyclohexane is that hexane is an acyclic alkane while cyclohexane is a cyclic alkane with a ring structure. They both have six carbon atoms, but a different number of hydrogen atoms.
How do you identify cyclohexane axial and equatorial?
- the equatorial bonds will form an “equator” around the ring.
- The axial bonds will either face towards you or away. These will alternate with each axial bond. The first axial bond will be coming towards with the next going away. There will be three of each type.
What is ring flipping in cyclohexane?
Ring flip (chair flip): The conversion of one cyclohexane chair conformation into another, by rotation around single bonds. Cyclohexane ring flip causes axial substituents to become equatorial, and equatorial substituents to become axial.
Which conformation of cyclohexane is hylian stable?
The stability order of conformation of cyclohexane is chair gttwist boat gt boat gt half chair . Half – Chair is lesss stable due to torsional and angle strain.
Which one of the following is the most energetic conformation of cyclohexane?
Most stable and least energetic conformation of cyclohexane is half-chair.
Which of the following cyclohexane conformations has the most energy is the least stable )? A chair B half chair C Boat D twist boat?
Boat conformation is the least stable, with the highest energy, has steric hindrance between the two equatorial hydrogens on carbon 1 and carbon 4, and has torsional tension because each bond almost completely ellipses other bonds in the Newman projection.
What makes a conformation less stable?
In an eclipsed conformation the carbons are aligned so that the hydrogens are lined up with each other. This creates steric hindrance between them. In a staggered conformation the atoms are all equally spaced from each other. … The eclipsed conformation of ethane is less stable than the staggered conformation.
Which one of the following conformation of cyclohexane is chiral?
Twist-boat conformation of cyclohexane is chiral.
Which of the following is the least energy conformer of cyclohexane?
Half chair conformer is least stable due to maximum strain.
Which is the following is most stable?
- A. Sn2+
- B. Ge2+
- C. Si2+
- D. Pb2+
- Due to inert pair effect which increases on moving down the group, Pb2+ is the most stable.
Which statement about cyclohexane is incorrect Mcq?
Which statement about cyclohexane is incorrect? Each C atom is sp3 hybridized. Cyclohexane suffers ring strain. The cyclohexane ring can flip between chair and boat conformers.
Which one of the following is most stable Mcq?
3∘ carbocations are most stable.
Which is more stable between cyclohexane or benzene?
Lower the heat of formation, more stable is the compound. From the above graph, it’s very clear that cyclohexane is more stable compared to benzene. Because cyclohexane has 50 kcals/mol less energy compared to benzene. As we know lower the heat of formation more stable the compound.
How can you distinguish between cyclohexane and cyclohexene using IR?
The major difference is that cyclohexene will possess a medium stretch anywhere between 1550–1650, whereas cyclohexane will not. Although I did not mention many of the other peaks that differ between the structures, you should still have no problem distinguishing between the two.
Which one is more stable benzene or cyclohexane?
Benzene is more stable than cyclohexane. The reason is cyclic conjugated dienes (alternate single and double bonds) are more stable due to resonance and while cyclohexane is not stablised by resonance due to which it is less stable.
Why is cyclohexane safer than hexane?
Density – the ring structure of cyclohexane gives it a higher density compared to the linear chain structure that n-hexane has. Toxicity – there are several reasons as to why n-hexane is more hazardous to work with than cyclohexane. The relatively high vapor pressure and known neurotoxicity of n-hexane are two examples.
Why does cyclohexane have a higher boiling point than cyclohexene?
Cyclohexene is planar, and can pack closer together which means more London Dispersion forces (van der waals(sp?) to us oldsters) holding them together making it take more energy to separate them, and by extension, a higher boiling point. Cyclohexane has all those axial hydrogens poking out.