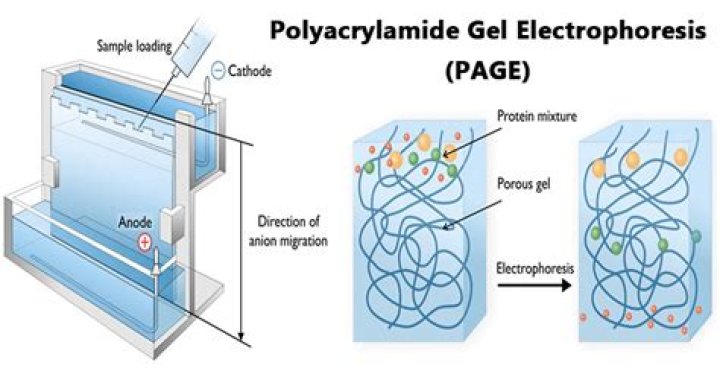
Polyacrylamide and agarose are two support matrices commonly used in electrophoresis. … Agarose has a large pore size and is suitable for separating nucleic acids and large protein complexes. Polyacrylamide has a smaller pore size and is ideal for separating majority of proteins and smaller nucleic acids.
Why is polyacrylamide used instead of agarose?
Agarose gels are used with DNA, due to the larger size of the biomolecules (DNA fragments are often thousands of kDa). For protein gels, polyacrylamide gives good resolution, as the far smaller size (50 kDa is typical) is more suited for the tighter intermolecular gaps of the gel.
What are the advantages of using polyacrylamide matrix?
- Stable chemically cross-linked gel.
- Greater resolving power (Sharp bands)
- Can accommodate larger quantities of DNA without significant loss in resolution.
- The DNA recovered from polyacrylamide gels is extremely pure.
Why polyacrylamide gel is used for protein separation?
Polyacrylamide gels are based on the free radical polymerization principle of acrylamide and cross-linking N,N′-methylene-bis-acrylamide. This material is physically very stable and strong. It is especially used for the electrophoretic separation of small or medium sized (up to about 1×106 Da) proteins.How does polyacrylamide gel electrophoresis separate proteins?
In PAGE, an anionic detergent called sodium dodecyl sulfate (SDS) is used to bind to proteins and give them a negative charge. Proteins are then separated electrophoretically according to their size using a gel matrix made of polyacrylamide in an electric field. … The more the cross-linking the harder the gel.
What is polyacrylamide used for?
High molecular weight (106–3 × 107 Da) polyacrylamide (PAM) is commonly used as a flocculant in water and wastewater treatment, as a soil conditioner, and as a viscosity modifier and friction reducer in both enhanced oil recovery and high volume hydraulic fracturing.
Why is polyacrylamide used for protein electrophoresis instead of agarose?
Polyacrylamide and agarose are two support matrices commonly used in electrophoresis. … Agarose has a large pore size and is suitable for separating nucleic acids and large protein complexes. Polyacrylamide has a smaller pore size and is ideal for separating majority of proteins and smaller nucleic acids.
Which of the following can SDS polyacrylamide electrophoresis be used to do?
Sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE) is commonly used to obtain high resolution separation of complex mixtures of proteins. The method initially denatures the proteins that will undergo electrophoresis.What is the principle of polyacrylamide gel electrophoresis?
PAGE (Polyacrylamide Gel Electrophoresis), is an analytical method used to separate components of a protein mixture based on their size. The technique is based upon the principle that a charged molecule will migrate in an electric field towards an electrode with opposite sign.
What is the role of SDS in protein electrophoresis?SDS is a strong detergent and present in high concentrations in the buffer that prepares samples for electrophoresis. … Before samples such as cells can be run on a protein gel, SDS needs to lyse cell membranes and solubilize all proteins.
Article first time published onWhy is protein electrophoresis done vertically?
The vertical system allows you to make them sequentially. You add the resolving gel first and then once it is set, you add the stacking gel. … Sandwiching it between two plates keeps oxygen away from the gel mix. So in an open, horizontal system the polymerization reaction would not proceed efficiently.
Which support matrix is used in the electrophoresis of proteins?
Polyacrylamide and agarose are two support matrices commonly used in electrophoresis. These matrices serve as porous media and behave like a molecular sieve. Agarose has a large pore size and is suitable for separating nucleic acids and large protein complexes.
What does polyacrylamide gel electrophoresis allow for when working with DNA?
Agarose gels can be used to resolve large fragments of DNA. Polyacrylamide gels are used to separate shorter nucleic acids, generally in the range of 1−1000 base pairs, based on the concentration used (Figure 1). … Native gels allow the DNA or RNA to remain double stranded.
What role does sodium dodecyl sulfate SDS in 2 dimensional electrophoresis?
Before separating the proteins by mass, they are treated with sodium dodecyl sulfate (SDS) along with other reagents (SDS-PAGE in 1-D). This denatures the proteins (that is, it unfolds them into long, straight molecules) and binds a number of SDS molecules roughly proportional to the protein’s length.
Why is agarose used for DNA gel electrophoresis?
Agarose permit the formation of bigger pores and can be used to solve bigger molecule as dna while acrylammide has smaller pores and it is able to solve small molecule as dna fragments or proteins. therefore two molecules with so different size need gels with different resolution.
What is sodium dodecyl sulphate SDS?
Sodium dodecyl sulfate (SDS) is an anionic detergent used to denature secondary and tertiary structure of the proteins (Shevchenko, Wilm, Vorm, & Mann, 1996). From: Advances in Protein Chemistry and Structural Biology, 2021.
Why polyacrylamide is not used in gel electrophoresis?
Increased concentrations of acrylamide result in decreased pore size after polymerization. Polyacrylamide gel with small pores helps to examine smaller molecules better since the small molecules can enter the pores and travel through the gel while large molecules get trapped at the pore openings.
How does agarose gel electrophoresis differ from polyacrylamide gel electrophoresis?
The main difference between agarose and polyacrylamide is that agarose is used in the agarose gel electrophoresis (AGE) mainly for the separation of DNA, whereas polyacrylamide is used in the polyacrylamide gel electrophoresis (PAGE) mainly for the separation of proteins.
What is an advantage of agarose over polyacrylamide gels quizlet?
Agarose polymer spaces are bigger than those of polyacrylamide. What is an advantage of agarose over polyacrylamide gels? Agarose monomers are not toxic. A very limited amount of nucleic acid, 500-1500 bp in size, is to be analyzed in a short time (same day) with the results available immediately.
Why is polyacrylamide used in water treatment?
Polyacrylamide is used to coagulate or flocculate solids in a liquid. This makes them useful in water treatment applications. Polyacrylamide increases the viscosity of water and is widely used in petroleum applications across the world to enhance oil recovery.
What are the properties of polyacrylamide?
Polyacrylamide (PAM) is a water-soluble synthetic polymer which possesses useful properties such as good adhesiveness, proper hygroscopicity, high hydrophilicity, and non-toxicity. PAM composites with CNCs can be obtained by mixing an aqueous solution of PAM and an aqueous suspension of CNCs.
Why polyacrylamide is soluble in water?
sightwhite, solid crystalssolubility in water215 g/ 100 ml at 30 deg C
What is the constituent of polyacrylamide gel?
Polyacrylamide gels are prepared by free radical polymerization of acrylamide and a comonomer crosslinker such as bis-acrylamide. Polymerization is initiated by ammonium persulfate (APS) with tetramethylethylenediamine (TEMED) as the catalyst (see figure below).
What is the role of SDS in SDS PAGE?
SDS (sodium dodecyl sulfate) is an anionic detergent that unfolds and denatures proteins, coating proteins in negative charge. It is added in excess to the proteins, so that the proteins’ intrinsic charge is covered, and a similar charge-to-mass ratio is obtained for all proteins.
What is the role of SDS in SDS PAGE Mcq?
Sodium dodecyl sulfate (SDS) is an anionic detergent and is most commonly used in protein chemistry to denature proteins and dissolve proteins and protein aggregates immersed in water, such as in SDS-PAGE. … It denatures proteins by binding to the protein chain with its hydrocarbon tail.
What is the function of 5% stacking gel in PolyacrylAmide gel electrophoresis?
The purpose of stacking gel is to line up all the protein samples loaded on the gel, so that they can enter the resolving gel at the same time.
Why SDS is used in DNA extraction?
SDS provides a negative charge to each protein as a function of their size. Accordingly, all of proteins have the same shape in the gel separation they are separated only for their size. Furthermore, SDS can be used to aid in lysing cell during DNA extraction.
What is the advantage of adding SDS to gel electrophoresis?
The advantage of adding SDS to gel electrophoresis is that it denatures the proteins and gives them a negative charge.
What is the difference between vertical and horizontal electrophoresis?
One of the key differences between the two systems is their orientation. In horizontal gel electrophoresis, the gel matrix is cast horizontally and submerged in a continuous running buffer while in vertical gel electrophoresis, the gel is vertically oriented and the buffer system is discontinuous.
What is the principle of vertical gel electrophoresis?
Vertical gel electrophoresis contains stacking gel and resolving gel. The stacking gel concentrates proteins that are loaded into the well so that the proteins can start to migrate at the same time. After stacking, the resolution gel separate proteins based on the molecular size.
Can a polyacrylamide gel run horizontally?
Thus excluding oxygen is important for gel polymerization, but as long as oxygen is excluded, the gel can be successfully poured in a horizontal position. I once saw a graduate student pour a polyacrylamide gel horizontally on one plate as is done with agarose gels.