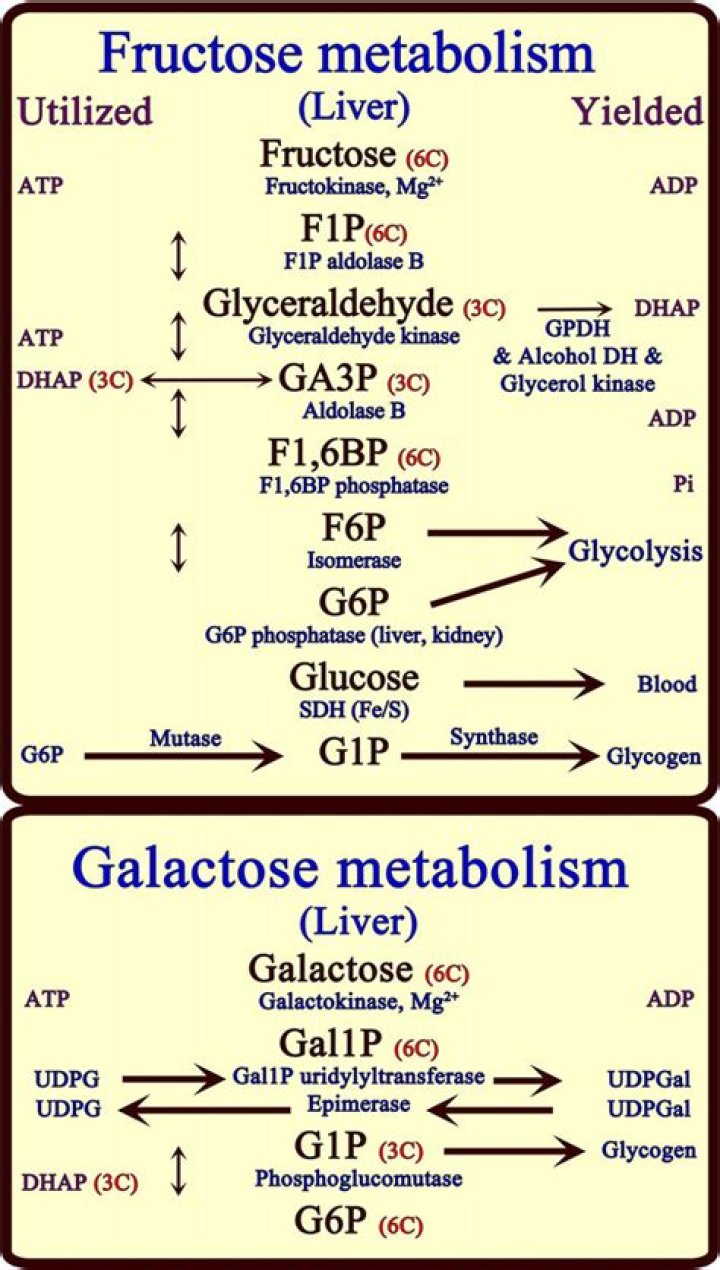
Glucose and galactose are stereoisomers (have atoms bonded together in the same order, but differently arranged in space). They differ in their stereochemistry at carbon 4. Fructose is a structural isomer of glucose and galactose (has the same atoms, but bonded together in a different order).
What is the term used to describe the structural relationship among glucose galactose and fructose?
Glucose, galactose, and fructose are monosaccharide isomers, which means they all have the same chemical formula but differ structurally and chemically. Disaccharides form when two monosaccharides undergo a dehydration reaction (a condensation reaction); they are held together by a covalent bond.
What is the relationship between glucose fructose and galactose quizlet?
fructose is absorbed by facilitated diffusion and glucose and galactose are absorbed by active transport. What happens to fructose and galactose once it enters the bloodstream? they are converted into glucose or used for energy.
What term describes the relationship between glucose and fructose?
Glycosidic bonds (also called glycosidic linkages) can be of the alpha or the beta type. Disaccharides: Sucrose is formed when a monomer of glucose and a monomer of fructose are joined in a dehydration reaction to form a glycosidic bond.What is the relationship between galactose and glucose?
Galactose is a monosaccharide and has the same chemical formula as glucose, i.e., C6H12O6. It is similar to glucose in its structure, differing only in the position of one hydroxyl group. This difference, however, gives galactose different chemical and biochemical properties to glucose.
What bond holds glucose together?
Tiny forces called hydrogen bonds hold the glucose molecules together, and the chains in close proximity. Although each hydrogen bond is very, very weak, when thousands or millions of them form between two cellulose molecules the result is a very stable, very strong complex that has enormous strength.
What are the structural isomers of glucose?
Glucose and its isomers Fructose is a structural isomer of glucose and galactose, meaning that its atoms are actually bonded together in a different order. Glucose and galactose are stereoisomers (have atoms bonded together in the same order, but differently arranged in space).
Are glucose and fructose functional isomers?
Thus, glucose and fructose have the same molecular formula with different functional groups and hence its an example of functional isomerism. Therefore, we can say that glucose and fructose are functional isomers.What is the major structural difference between starch and cellulose?
Starch is formed from alpha glucose, while cellulose is made of beta glucose. The difference in the linkages lends to differences in 3-D structure and function. Starch can be straight or branched and is used as energy storage for plants because it can form compact structures and is easily broken down.
Are glucose and fructose Anomers?An anomer is a distinct type of epimer in which one of two stereoisomers of a cyclic saccharide differs only in its configuration at the acetal carbon, also called the anomeric carbon. Glucose and fructose are not anomers.
Article first time published onWhat is the relationship between glucose fructose mannose and galactose?
Galactose and mannose are epimers of the glucose molecule. The key difference between glucose galactose and mannose is that glucose is a six-carbon structure and galactose is the C4 epimer of glucose whereas mannose is the C2 epimer of glucose. Moreover, glucose is naturally produced through photosynthesis in plants.
Which molecule breaks the bond between glucose and fructose?
Explanation: The bonds between the glucose and fructose molecules making up the disaccharide sucrose break due to the enzyme sucrase, and water is added as H and OH to the two molecules. This is called a hydrolysis reaction.
How are glucose and galactose absorbed quizlet?
Glucose and galactose are absorbed by active transport (requires more energy) in the small intestine, and fructose is absorbed by facilitated diffusion in the small intestine.
What is the structural difference between glucose and fructose?
Ans: Glucose is a 6 membered ring, whereas the Fructose is 5 membered ring. Glucose produces less fat compared to fructose in our body. Glucose is an aldohexose, whereas the Fructose is a Ketohexose.
What is the structure of glucose and fructose?
Glucose and fructose are functional isomers of each other because they have same molecular formula that is C6H12O6 But different functional group in their chemical formula. Glucose has aldehyde group while fructose has ketone as functional group. They differ in the nature of the functional group.
What is the structural relationship between the common monosaccharides D glucose and D galactose?
Both monosaccharides are D-sugars, meaning that the -OH group on carbon-5 of these hexoses is located on the right in Fischer Projection. The only difference between D-glucose and D-galactose is on carbon-4. For D-glucose, the -OH is on the right in Fischer Projection, and for D-galactose, the -OH group is on the left.
Why are glucose and fructose structural isomers?
For Example, fructose and glucose are structural isomers because, although they have the same molecular formula (C6H12O6), glucose contains an aldehyde group (it is an aldose) and fructose contains a keto group (it is a ketose).
Are galactose and fructose isomers?
Notice that glucose, galactose, and fructose all have the same molecular formula, C6H12O6. They are isomers.
Are glucose and galactose geometric isomers?
Glucose and galactose differ in the configuration at C-4 , they are non-superimposable, and they are not mirror images of each other.. They are therefore diastereoisomers.
How do glucose molecules join together to form polysaccharides?
Glucose molecules can link together. … When many glucose molecules link together in a long chain, molecules called polysaccharides (many sugars) are formed. The link formed between each molecule is known as a glycosidic bond. Amylose has several hundred glucose monomers linked together in this way.
What is the name of the chemical formed when glucose molecules join together?
Two glucose molecules react to form the disaccharide maltose. Starch and cellulose are polysaccharides made up of glucose units. Galactose molecules look very similar to glucose molecules. They can also exist in α and β forms.
What forms after glycosidic linkage between glucose and galactose is complete?
Lactose is composed of a molecule of galactose joined to a molecule of glucose by a β-1,4-glycosidic linkage. It is a reducing sugar that is found in milk. Sucrose is composed of a molecule of glucose joined to a molecule of fructose by an α-1,β-2-glycosidic linkage.
What is the basic structural difference between starch and glucose?
Starch contains glucose residues as α(1-4) glycosidic bonds in amylose, while glycosidic bonds at branching points in amylopectin α(1-6), otherwise α(1-4) bonds. Cellulose constitutes their residues of glucose as glycosidic bonds with β(1-4).
How is glucose different from cellulose in terms of structure?
1. Glucose is made from the process of photosynthesis while cellulose is made from many chains of glucose after glucose is dissolved as energy and stored as starch. … Glucose is considered as a simple sugar while cellulose is a complex carbohydrate.
What is the main structural difference between starch and cellulose quizlet?
What is the main structural difference between starch and cellulose? The way their glucose units are hooked together. Starch is arranged in alpha linkage (Oxygen atom connecting glucose units is pointed down) and cellulose is arranged in beta linkage (Oxygen atom connecting glucose units are pointed up).
What type of isomerism exist between glucose and galactose?
Diastereomers are optical isomers of each other with not being mirror images of each other. Since the same can be seen in case of glucose and galactose, hence they are diastereomers. -Therefore, glucose and galactose are diastereomers.
Do structural isomers have the same functional group?
In this variety of structural isomerism, the isomers contain different functional groups – that is, they belong to different families of compounds (different homologous series). A molecular formula C3H6O could be either propanal (an aldehyde) or propanone (a ketone).
Are glucose and fructose tautomers?
Glucose and fructose are chain isomers.
Is glucose and galactose are Anomers?
Glucose and galactose differ at C-4 position and hence are C4 epimers.
Is glucose and galactose are epimers?
Hint: Carbohydrates that differ in the location of the hydroxyl (i.e., -OH) group only at one position are known as epimers. So, glucose and galactose are epimers as they have identical configuration at all the positions except for one position.
What term is used to describe the relationship between galactose and mannose?
Galactose and Mannose are epimers of glucose. Epimers are compounds having the same chemical formula but differ in the spatial arrangement around a single carbon atom.