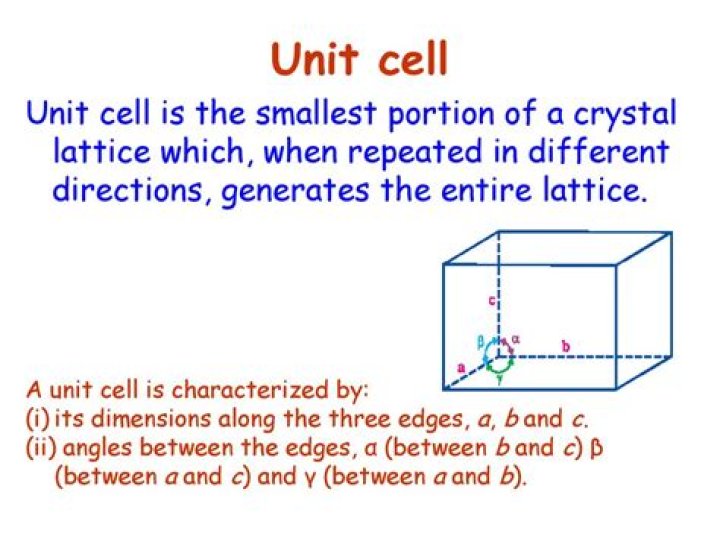
The unit cell is defined as the smallest repeating unit having the full symmetry of the crystal structure. The geometry of the unit cell is defined as a parallelepiped, providing six lattice parameters taken as the lengths of the cell edges (a, b, c) and the angles between them (α, β, γ).
What is unit cell in simple words?
Definition of unit cell : the simplest polyhedron that embodies all the structural characteristics of and by indefinite repetition makes up the lattice of a crystal.
How do you find the unit cell of a crystal?
The Simplest Repeating Unit in a CrystalA Three-Dimensional GraphDetermining the Unit Cell of a CrystalCalculating Metallic or Ionic Radii
What is unit cell and lattice of a crystal?
Unit Cell is the smallest part (portion) of a crystal lattice. It is the simplest repeating unit in a crystal structure. The entire lattice is generated by the repetition of the unit cell in different directions.What is unit cell used for?
A unit cell is the most basic and least volume consuming repeating structure of any solid. It is used to visually simplify the crystalline patterns solids arrange themselves in. When the unit cell repeats itself, the network is called a lattice.
What is a unit cell in chemistry class 12?
Unit cell can be described as the smallest portion of a crystal lattice. A unit cell is characterized by: (i) Its dimensions along the three edges, a, b and c which may or may not be mutually perpendicular. … Thus, a unit cell is characterized by six parameters a, b, c, α, β and γ.
What is unit cell in chemistry in one word?
unit cell – the smallest group of atoms or molecules whose repetition at regular intervals in three dimensions produces the lattices of a crystal.
How many unit cells are present in a crystal lattice?
∴ Unit cell =1.029×10224=2.57×1021 unit cell.How do you find the unit cell?
We measure up the mass of one unit cell to the mass of all the atoms present in that same cell. The number of atoms is going to depend on the type of cell it is. So we multiply the number of atoms “n” into the mass of each atom “m” to achieve the mass of a unit cell.
What is unit cell and primitive cell?A unit cell is the smallest group of atoms which has the overall symmetry of a crystal, and from which the entire lattice can be built up by repetition in three dimensions. A primitive cell in chemistry is the smallest possible unit cell of a lattice, having lattice points at each of its eight vertices only.
Article first time published onWhat is unit cell class 9?
The smallest repeating unit of the crystal lattice is the unit cell, the building block of a crystal. The unit cells which are all identical are defined in such a way that they fill space without overlapping.
What is unit cell explain the types of unit cell?
Answer: A unit cell is the smallest repeating portion of a crystal lattice. Unit cells occur in many different varieties. As one example, the cubic crystal system is composed of three different types of unit cells: (1) simple cubic , (2) face-centered cubic , and (3)body-centered cubic .
What are the types of unit cell?
There are three types of unit cells present in nature, primitive cubic, body-centered cubic, and face-centered cubic.
What is a unit cell in physics?
In geometry, biology, mineralogy, and solid state physics, a unit cell is a repeating unit formed by the vectors spanning the points of a lattice.
What is the unit cell constant?
The cell constant, known as K, refers to a theoretical electrode consisting of two 1 cm square plates 1 cm apart. A cell constant has units of 1/cm (per centimeter), where the number refers to the ratio of the distance between the electrode plates to the surface area of the plate.
What is a crystal lattice in chemistry?
Definition of crystal lattice : the arrangement of atoms, molecules, or ions of a crystal in the form of a space lattice.
What is the size of unit cell of the Aluminium crystal?
The unit cell of aluminium is cube with an edge length of 405 pm.
How many atoms are in a unit cell?
The smallest repeating unit of a crystal lattice is the unit cell. The simple cubic unit cell contains only eight atoms, molecules, or ions at the corners of a cube. A body-centered cubic (bcc) unit cell contains one additional component in the center of the cube.
What is unit cell in chemistry Shaalaa?
Solution. A basic repeating structural unit of a crystalline solid is called a unit cell.
What is unit cell in chemistry class 12 shaala?
The smallest repeating structural unit of a crystalline solid is called unit cell.
What is unit cell 12th HSC?
A unit cell is the smallest repeating structural unit of a crystalline solid. … A crystalline solid can be obtained by repeating the unit cells (of the same crystalline substance) in space in all directions. ii.
How many atoms are in a unit cell in Gaas crystal structure?
There are 14 gallium atoms shown, but only 4 arsenic atoms. However, given that 8 cells share each corner atom and 2 cells share each face atom, it is seen that there are actually 4 arsenic and 4 gallium atoms per cubic unit cell, i.e. 4 basis molecules per cell. 1.
How many unit cells are present in 5g of crystal AB?
In 5.0g of crystal AB there are 540×NA 5 40 × N A molecules and 540×NA×14 5 40 × N A × 1 4 unit cells.
How many units are present in cube shaped?
48×1021 unit cells.
How many molecules are there in the unit cell of NaCl?
So, each unit cell has 4 atoms of each Na+ and Cl-. Hence there are total 4 molecules of NaCl in each unit cell.
What is primitive cell in crystal structure?
A primitive cell is a unit cell that contains exactly one lattice point. It is the smallest possible cell. If there is a lattice point at the edge of a cell and thus shared with another cell, it is only counted half. Accordingly, a point located on the corner of a cube is shared by 8 cubes and would count with 18 .
What is primitive cell in chemistry?
A primitive cell is a unit cell built on the basis vectors of a primitive basis of the direct lattice, namely a crystallographic basis of the vector lattice L such that every lattice vector t of L may be obtained as an integral linear combination of the basis vectors, a, b, c.
Are all primitive cells unit cells?
No,not all unit cells are primitive. Non-primitive unit cells, however, contain extra lattice points not at the corners.
Which type of crystal is a cubic crystal?
SilicidesGermanidesChromiumChromium(IV) silicideChromium(IV) germanide
What are the four different types of unit cells?
rhombohedral, hexagonal, triclinic – one unique form each. tetragonal – simple and body-centered. monoclinic – simple and base-centered. orthorhombic – simple, face-centered, body-centered, base-centered.
What are the characteristics of unit cell?
- A unit cell has three edges a, b and c and three angles α, β and γ between the respective edges.
- The a, b and c may or may not be mutually perpendicular.
- The angle between edge b and c is α, a and c is β and that of between a and b is γ