Ethene is an symmetrical alkene with two carbon atoms. With HBr, ethene readily reacts and give ethyl bromide (bromoethane) as the product.
What happens when ethene reacts with hydrogen?
The hydrogenation of ethene Ethene reacts with hydrogen in the presence of a finely divided nickel catalyst at a temperature of about 150°C. Ethane is produced. This is a fairly pointless reaction because ethene is a far more useful compound than ethane!
What is the product of the reaction of hydrogen chloride and ethene?
On reacting ethene with HCl we get chloroethane. It is an addition reaction.
What is the product of ethene and hydrogen?
Alkenes react with hydrogen bromide in the cold. The double bond breaks and a hydrogen atom ends up attached to one of the carbons and a bromine atom to the other. In the case of ethene, bromoethane is formed.What is the product of an alkene and hydrogen?
An example of an alkene addition reaction is a process called hydrogenation.In a hydrogenation reaction, two hydrogen atoms are added across the double bond of an alkene, resulting in a saturated alkane.
What substance is added to ethene to ethane?
This process is called catalytic hydrogenation as it involves a catalyst to drive the reaction and produces alkanes as a product. In the given scenario, as ethene ( ) is an alkene, it can be converted into ethane by catalytic hydrogenation or Addition of hydrogen.
What is the product of ethene and bromine?
The electrophilic addition of bromine to ethene The bromine loses its original red-brown color to give a colorless liquid. In the case of the reaction with ethene, 1,2-dibromoethane is formed. This decoloration of bromine is often used as a test for a carbon-carbon double bond.
What happens when ethene reacts with hydrogen bromide?
when you mix ethene (a member of alkene group) and hydrogen bromide . … The double bond breaks and a hydrogen atom ends up attached to one of the carbons and a bromine atom to the other. In the case of ethene , bromoethane is formed.What is the product of the reaction between ethene and hydrogen bromide?
Ethene is an symmetrical alkene with two carbon atoms. With HBr, ethene readily reacts and give ethyl bromide (bromoethane) as the product.
What is ethene formula?Ethylene, or ethene, is an unsaturated hydrocarbon. It is a colorless gas. Its chemical formula is C2H4 where there is a double bond between the carbons.
Article first time published onWhat product is formed when ethene reacts with chlorine?
Ethene and Chlorine or Bromine or Iodine For example, bromine adds to give 1,2-dibromoethane.
Which word describes the reactions of ethene with hydrogen and chlorine?
This is called hydrogenation , and it needs a catalyst .
What are the products of an addition reaction with HBr?
HBr adds to alkenes to create alkyl halides. A good way to think of the reaction is that the pi bond of the alkene acts as a weak nucleophile and reacts with the electrophilic proton of HBr.
What is the order of hydrogenation of ethene?
The reaction is first order with activation energy 15.8 kJ mol−1. With an equal volume of HZ, the alkene is 100% catalytically hydrogenated to C2H6 by a first-order process with activation energy 45.5 kJ mol−1. Increasing the proportion of H2 increases the rate of catalysis.
What is hydration of ethene?
Example of Hydration of Alkene: Hydration of ethene (ethylene) to ethanol. … The active site on the ethene molecule is the double bond (C=C). In the presence of a dilute strong acid, water will add across the double bond in ethene (ethylene) to produce ethanol (ethyl alcohol).
Why does ethene not react with iodine?
Alkenes contain at least one carbon-carbon double bond. … Remember the reactivity of halogens decreases down the group, therefore the addition reaction of alkenes with chlorine takes place faster with chlorine than for bromine with iodine being the slowest.
What type of reaction is ethene and bromine?
Ethene and bromine are an addition reaction because ethene is an alkene – it has a double bond. It is easier for new atoms to open the double bond and react there than to remove the hydrogen already attached, and then bond to it, which would be a substitution reaction.
What is the chemical reaction of ethene?
Reactions of Ethene (ethylene) Ethene (ethylene) undergoes complete combustion in excess oxygen to produce carbon dioxide and water. Ethene (ethylene) undergoes addition reactions with: halogens. haloalkanes (alkyl halides)
What happens when bromine reacts with Cyclopropane?
In the absence of UV light, cyclopropane can undergo addition reactions in which the ring is broken. For example, with bromine, cyclopropane gives 1,3-dibromopropane. This can still happen in the presence of light – but you will get substitution reactions as well.
How is ethane prepared from ethene?
Ethane when reacted with Chlorine (halogenation) in presence of sunlight gives ethyl chloride. Now, Ethyl chloride on reaction with KOH (Alcoholic) gives ethene.
How do ethane and ethene react?
Ethene undergoes addition reactions, due to its double bonds. For example, in the hydrogenation reaction, two hydrogens are added to the double bond and converting ethene to ethane.
What type of reaction is ethene to ethanol?
In a hydration reaction, the water molecule reacts with an alkene to generate an alcohol. Ethanol can be prepared by the reaction of alkene with the water molecule. Ethene molecules react with sulphuric acid generating an alcohol, that is ethanol.
Which product is obtained by reaction between 2 methyl propane and HBr?
1-bromo-2-methyl propane.
How does ethene gas react with halogen acid?
(ii) When vapours of ethene and hydrobromic acid are mixed at room temperature, they react to form addition product, bromoethane. … (vi) Ethene reacts with HCl to form ethane glycol monochloride.
What do you mean by addition reaction?
addition reaction, any of a class of chemical reactions in which an atom or group of atoms is added to a molecule. … An addition reaction may be visualized as a process by which the double or triple bonds are fully or partially broken in order to accommodate additional atoms or groups of atoms in the molecule.
What happens when ethene reacts with alkaline kmno4?
When ethylene or ethene reacts with alkaline potassium permanganate it gives ethane-1,2-diol or ethylene glycol.
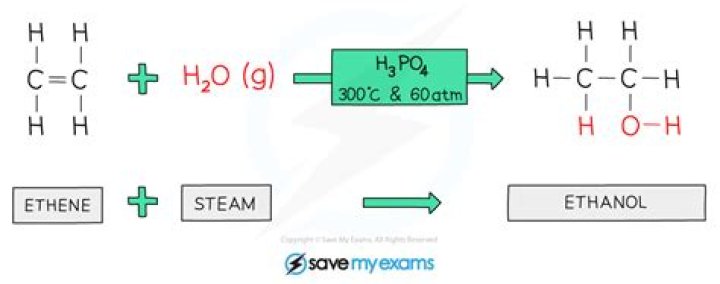
What happens when ethene reacts with water?
Alkenes undergo an addition reaction with water in the presence of a catalyst to form an alcohol. … In this process ethene and steam (water in the gaseous phase) are passed at 300°C and a pressure approximately 60 times above atmospheric pressure over a phosphoric acid catalyst to produce ethanol.
How is bromoethane prepared from ethene?
Ethane is converted to bromoethene by first reacting ethane with bromine in presence of U.V light to form bromoethane. Then bromoethane is reacted with alcoholic potassium hydroxide to form ethene.
Are ethene and ethylene the same?
Ethylene is the most produce compound in organic chemistry. Ethylene (also called Ethene; C2H4), the simplest Alkene, is an organic compound containing a C=C double bond. Ethylene is a coplanary unsaturated hydrocarbon (also called olefin) which is the most produced for industrial use.
What is the chemical name of ch2?
PubChem CID123164Molecular FormulaCH2SynonymsMethylene 2465-56-7 methanediyl singlet carbene triplet carbene More…Molecular Weight14.027DatesModify 2022-01-08 Create 2004-09-16
How many bonds does ethene have?
Ethene is not a very complicated molecule. It contains two carbon atoms that are double bonded to each other, with each of these atoms also bonded to two Hydrogen atoms. This forms a total of three bonds to each carbon atom, giving them an sp2 hybridization.