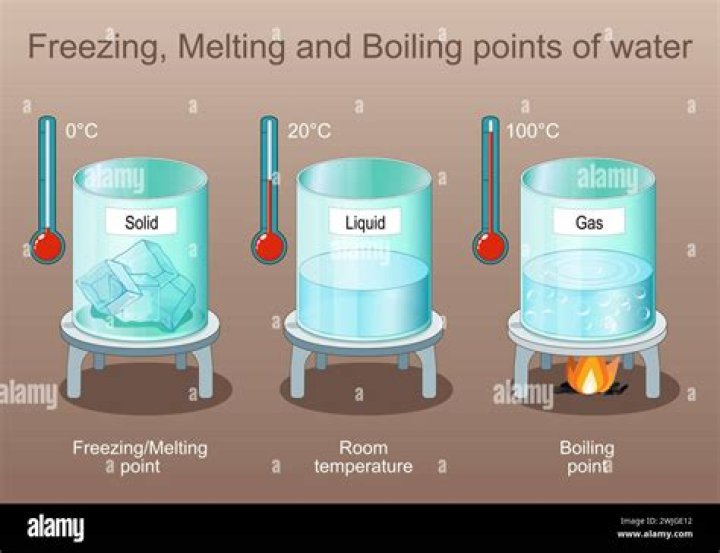
Physical changes occur in a substance during melting, which is the process of turning a solid into a liquid, and freezing, which is the process of turning a liquid into a solid. … For example, melting requires the addition of heat to a substance, while freezing occurs with the removal of heat.
What is the process of freezing?
Freezing is the process that causes a substance to change from a liquid to a solid. Freezing occurs when the molecules of a liquid slow down enough that their attractions cause them to arrange themselves into fixed positions as a solid.
Are freezing and melting reverse process?
Freezing is the process of changing a liquid into a solid by cooling whereas melting is the process of changing a solid into a liquid by heating. Hence it can be said that freezing is the reverse of melting.
What is the process of melting?
Melting is a process that causes a substance to change from a solid to a liquid. Melting occurs when the molecules of a solid speed up enough that the motion overcomes the attractions so that the molecules can move past each other as a liquid.What is the process of ice melting called?
Melting is also known as fusion, although this term has several meanings. Melting occurs when the internal energy of a solid increases, usually through the application of heat or pressure, such that the molecules become less ordered.
What is meant by freezing?
Freezing is a phase transition where a liquid turns into a solid when its temperature is lowered below its freezing point. In accordance with the internationally established definition, freezing means the solidification phase change of a liquid or the liquid content of a substance, usually due to cooling.
What is freezing and example?
Freezing is the process when a liquid turns into a solid. … One example of freezing is when water turns into ice. Freezing is the opposite of melting, and two steps away from evaporation. Freezing occurs at below 0 degrees Celsius with water, while some other liquids become a solid at higher or lower temperatures.
How are freezing and melting similar?
Similarities between melting and freezing Both melting and freezing are phase transition processes and cause change in state or phase of the substance. Both are physical change, so no new substance formation occurs. Both changes are reversible. Both greatly depend on external pressure on the substance.What is melting point of ice?
The melting point is the temperature at which a solid turns to a liquid. The melting point at which ice — a solid — turns to water — a liquid — is 32°F (0°C).
What is the meaning of melting of ice?1 to liquefy (a solid) or (of a solid) to become liquefied, as a result of the action of heat. 2 to become or make liquid; dissolve.
Article first time published onWhat is freezing and freezing point?
Freezing point is the temperature at which a liquid becomes a solid at normal atmospheric pressure. … The normal freezing point is the temperature at which a substance melts (or freezes) at one atmosphere (760 torr = 760 mm Hg = 14.7 psi = 101.3 kPa) of pressure.
Why is freezing and melting important?
This means that we can use the freezing or melting point as an indicator of the purity of a substance. When a solid is melted by heating or a liquid frozen while cooled, the temperature remains constant.
Why does melting and freezing occurs at the same temperature?
Melting and freezing of pure substances takes place at the same temperature since you are either breaking or forming the intermolecular forces that exist between molecules. Adding energy causes melting while removing energy forms bonds.
Why is melting called fusion?
Melting is called fusion because when two separate solid objects made from the same substance are melted, they can get mixed together into a new one (they fuse). This can also apply for different substances, like alloys from different metals or mixtures of alcohol and water, or different waxes and oils.
How are the processes of melting and freezing similar Quizizz?
How are the processes of melting and freezing similar? They are both processes that physically change a substance.
What are 3 examples of freezing?
- Snowfall. …
- Sea Ice. …
- Frozen Food. …
- Lava Hardening into Solid Rock. …
- Solidification of Melted Candle Wax. …
- Anti-freezer. …
- Embryo Freezing.
What is melting point?
melting point, temperature at which the solid and liquid forms of a pure substance can exist in equilibrium. As heat is applied to a solid, its temperature will increase until the melting point is reached.
What is freezing 4th class?
Solution. Ice is a solid form of water. At temperature below 0° C water becomes ice. This process is called freezing. Concept: Change in State of Water.
What is freezing point of liquid?
noun Physical Chemistry. the temperature at which a liquid freezes: The freezing point of water is 32°F, 0°C.
What is freezing cold?
Freezing is the process in which something changes from liquid to solid as the temperature becomes lower. … Freezing of the water system in cold weather is a problem in steam engines. Freezing is the process in which something changes from liquid to solid as the temperature becomes lower.
What is freezing and condensation?
Freezing: the substance changes from a liquid to a solid. Melting: the substance changes back from the solid to the liquid. Condensation: the substance changes from a gas to a liquid. … Sublimation: the substance changes directly from a solid to a gas without going through the liquid phase.
Why is freezing exothermic?
When water becomes a solid, it releases heat, warming up its surroundings. This makes freezing an exothermic reaction. … In order for water to change from a solid into a liquid, it needs to draw heat into itself from its surroundings.
What melts ice fastest?
Salt will always melt ice quicker than both of them. This is because in the same amount or volume, there are more molecules of salt than sugar or baking soda due to the chemical make-up. Salt, baking soda, and sugar will all act to lower the freezing point of the ice, making it melt quicker than the untouched ice cube.
What does salt do to ice?
Why does salt melt ice and snow? Salt causes a phenomenon called freezing point depression, which means it lowers the freezing point of water. … Just as salt lowers the freezing point of water in cold temperatures, it also raises the boiling point in hot temperatures.
What is lowest melting point?
The chemical element with the lowest melting point is Helium and the element with the highest melting point is Carbon. The unity used for the melting point is Celsius (C).
What is the process of ice turning into water?
When solid ice gains heat, it changes state from solid ice to liquid water in a process called melting. Ice cubes in a cold drink, for example, gradually melt. … This change, directly from a solid to a gas without becoming a liquid, is called sublimation.
What is the difference between heating and freezing?
When the heat of the solid is taken away, the heat inside the solid keeps on decreasing, which results in the solid change into a liquid. When the temperature decreases in the liquid, it starts forming solid. It completely forms solid when it reaches its freezing point.
Is melting and fusion same?
Answer) Melting is described as a process in which the solid changes the liquid state without even any change in temperature. This melting process is also known as a fusion process.
Is ice melting chemistry or physics?
Ice melts due to the chemical properties of water. There are more hydrogen bonds between the molecules of ice than in water. Ice begins to melt when its temperature exceeds 0 degrees Celsius and hydrogen bonds between water molecules break.
What is melting explain with an example?
The definition of melt is to turn from a solid to liquid as a result of exposure to heat. An example of melt is what an ice cube does when exposed to the sun. … An example of melt is the effect of adding ice or sugar to ice to lower the freezing point.
What is freezing and boiling point?
The Fahrenheit scale defines the freezing point of water as 32°F and the boiling point as 212°F. The Celsius scale sets the freezing point and boiling point of water at 0°C and 100°C respectively.