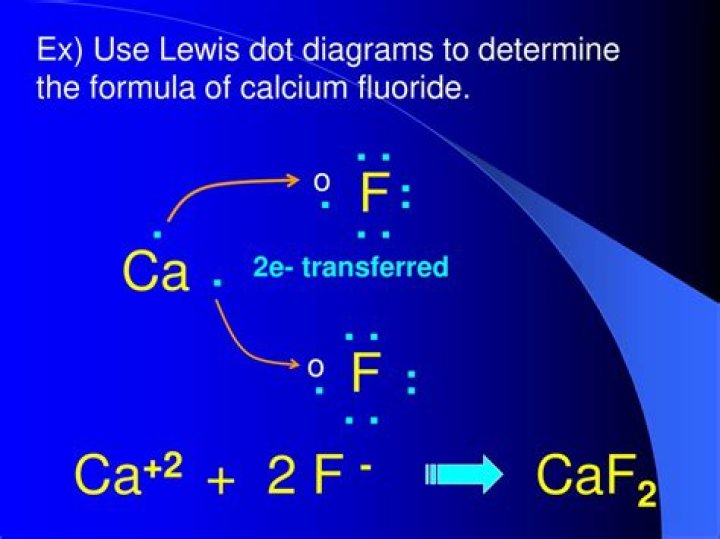
o The Ksp for CaF2 is 3.9 × 10–11 at 25 °C.
What is the KSP expression for CaF2?
o The Ksp for CaF2 is 3.9 × 10–11 at 25 °C.
What is the solubility of CaF2 in water?
The molar solubility of CaF2 in pure water is 3.32 x 10-4 mol/L.
What is the molar solubility of CaF2?
The molar solubility of CaF2 (Ksp = 5.3 × 10-11) in 0.1 M s.How KSP is calculated?
Next we write out the expression for Ksp , then “plug in” the concentrations to obtain the value for Ksp. Let’s do an example: The solubility of Ag2CrO4 in water is 1.31 x 10-4 moles/L. Calculate the value of Ksp . Using mole ratios, the [Ag+] will go up by (2 x 1.31 x 10-4 moles/L) = 2.62 x 10-4 moles/L.
What is the Ksp of BaF2?
The Ksp for BaF2 is 1.0 x 10^-6.
What is the Ksp of PbF2?
The Ksp for PbF2 is 4.0 x 10-8.
How can the solubility S of CaF2 be written in terms of solubility multiplication?
Solubility (s) of CaF_(2) in terms of its solubility product is given as. s=√Ksp4.How is molar solubility related to KSP?
A substance’s solubility product (Ksp) is the ratio of concentrations at equilibrium. Molar solubility, which is directly related to the solubility product, is the number of moles of the solute that can be dissolved per liter of solution before the solution becomes saturated.
Is CaF2 aqueous?Calcium fluoride is the inorganic compound of the elements calcium and fluorine with the formula CaF2. It is a white insoluble solid.
Article first time published onWhat is the solubility of CaF2 S in terms of molarity?
Question: What is the molar solubility of CaF2 in 0.10 M NaF solution 25 degrees C? The Ksp for CaF2 is 3.4×10-11. The answer is 3.4×10-9 M.
How do you calculate experimental KSP?
Ksp = [Ag+] [C2H3O2- ] (2) Equation (2) implies that in any system containing solid AgC2H3O2 (s) in equilibrium with its ions, the product of [Ag+] times [C2H3O2- ] will at a given tem perature have a fixed m agnitude , independent of how the equilibrium system was initially made up.
What is the KSP of AgCl?
CompoundCompoundAgCl1.8 × 10 -10Fe(OH) 2Al(OH) 33.0 × 10 -34Mg(OH) 2BaCO 35.0 × 10 -9PbCl 2BaSO 41.1 × 10 -10PbCO 3
What does PbF2 dissociate into?
Step 2: Solve. The dissociation equation shows that, for every mole of PbF2 that dissociates, 1 mol of Pb2+ and 2 mol of F− are produced.
When can KSP values be used to compare?
The relative MOLAR solubility of salts (saturated solution) can be determined by comparing Ksp values. The greater the Ksp the more ions are in solution, hence the greater the molar solubility. However, you can only directly compare salts that give equivalent numbers of ions in solution.
What is the KSP of CuSO4?
FormulaNamepKspCuSO4:5H2OChalcanthite2.64Fe(OH)2Fe(OH)214.08Fe(OH)3Fe(OH)3(a)37.08Fe2O3Hematite87.95
What is the KSP of CaSO4?
Calculate the solubility in g/L of calcium sulfate, CaSO4, given that its Ksp is 2.4 x 10-5. The dissolution of calcium sulfate forms equal amounts of calcium ions and sulfate ions according to the following equation.
What is the KSP of mnoh2?
The Ksp of manganese(II) hydroxide, MN(OH)2, is 2.00×10-13.
What does KSP depend on?
The solubility product is a kind of equilibrium constant and its value depends on temperature. Ksp usually increases with an increase in temperature due to increased solubility.
What is the molar solubility s of La IO3 3 in terms of the KSP?
The solubility of La(IO3)3 in a 0.62 M KIO3 solution is 1.0 x 10^-7 mol/L.
What is the molar solubility of pb3 po4 2?
The molar solubility of Pb3(PO4)2 in pure water is 6.2×10-12 M.
What is the solubility product of CaF2 if its saturated solution?
The solubility of calcium fluoride in saturated solution, it its solubility product is 3.2×10−11 is : 2.0×10−4M.
How many times is solubility of CaF2 decreased?
The more soluble substance means it has a higher solubility product value. Solubility product is only applicable to the sparingly soluble solution. Hence, 100 times solubility \[Ca{{F}_{2}}\] is decreased in $4\times {{10}^{-3}}M$ KF (aq) solution as compared to pure water at${{25}^{o}}C$.
What is the molar solubility of AgCl KSP 1.8 10 10?
This is the answer because there is a one-to-one relationship between the Ag+ dissolved and the AgCl it came from. So, the molar solubility of AgCl is 1.33 x 10¯5 moles per liter.
What is CaF2 in chemistry?
Calcium fluoride (CaF2) is the primary source of fluorine worldwide. It occurs as the mineral fluorite or fluorspar. Unlike the other calcium halides, it is practically insoluble in water.
What molecule is CaF2?
Calcium fluoride (CaF2) is an insoluble ionic compound composed of Ca2+ and F− ions. It occurs naturally as the mineral “Fluorite” (also called fluorspar) and as “Blue-John”. This salt is the source of most of the world’s fluorine.
What is the molar solubility of CaF2 in 0.1 M NAF?
3. 4×10−9M.
What is the solubility of CaF2 in pure water at 25?
The solubility of CaF2 in water at 25 °C is 1.7x10g per 100cc The solubility product of Caf, at 25 °C is about X x 10-11 mol/L.
Is KSP dependent on pH?
(1) CaF2 Ca2+ + 2F -Ksp = 3.45×10-11(2) HF H+ + F -Ka = 6.3×10-4
What does the value of KSP tell you in terms of equilibrium?
The solubility product constant (Ksp) describes the equilibrium between a solid and its constituent ions in a solution. The value of the constant identifies the degree to which the compound can dissociate in water. The higher the Ksp, the more soluble the compound is.