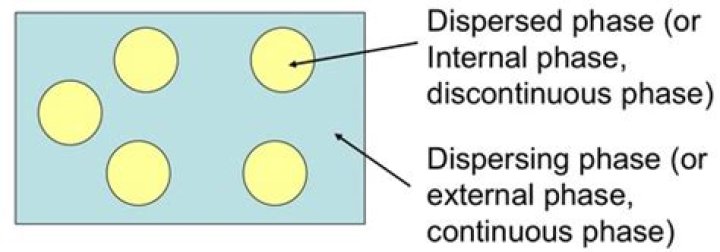
: the phase in a two-phase system that consists of finely divided particles (as colloidal particles), droplets, or bubbles of one substance distributed through another substance.
What is the Definition of dispersion medium?
Medical Definition of dispersion medium : the liquid, gaseous, or solid phase in a two-phase system in which the particles of the dispersed phase are distributed. — called also continuous phase, external phase.
What is the dispersed phase and dispersed medium?
The phase that is scattered or present in the form of colloidal particles is called dispersed phase and the medium in which the colloidal particles are dispersed is called the dispersion medium.In a starch solution ,starch represents the dispersed phase while water represents the dispersion medium.
What is a dispersed phase in a colloid?
The dispersed phase of dispersion is the discontinuous phase that distributes throughout the dispersion medium. It is one of the two stages of a colloid. The dispersion medium is the continuous phase of a colloid, and it distributes throughout the dispersion medium.What is continuous phase and dispersed phase?
The phase existing as small droplets is called the dispersed phase and the surrounding liquid is known as the continuous phase. Emulsions are commonly classified as oil-in-water (O/W) or water-in-oil (W/O) depending on whether the continuous phase is water or oil.
What is the dispersed phase in milk?
In the case of milk, milk is an emulsion in which both dispersed phase and dispersed medium are liquid states. In milk liquid fat is dispersed in water so that the fat is the dispersed phase and water represents the dispersion medium.
What is dispersed phase class 9?
Dispersed phase: The phase that is scattered or present in the form of colloidal particles is known as dispersed phase. Dispersion medium: The medium in which the colloidal particles are dispersed is called dispersion medium. … It is a collection of tiny solid, liquid and gas particles.
What is the difference between dispersed phase and dispersion phase?
A dispersion is a system in which particles of one phase are dispersed throughout a medium that is in a different phase. … The main difference between dispersed phase and dispersed medium is that dispersed phase is a discontinuous phase whereas dispersion medium is a continuous phase.What is the dispersed phase in a gel?
Gel is a system in which liquid is the dispersed phase and solid is the dispersion medium.
What is dispersed phase and dispersion medium of clouds?Cloud is an aerosol that means it is a colloidal suspension of water droplets in air. Hence, dispersed phase is liquid (droplets of water) and dispersion medium is gas (air).
Article first time published onWhat is the dispersed phase in blood?
ii) Blood: Dispersed phase is hormones, Respiratory gases, RBC, WBC, Platelets, proteins. And Dispersed medium is Plasma. iii) Printing ink: Dispersed phase is solid and dispersion medium is liquid.
What is emulsion phase separation?
Phase separation processes take place, where a liquid layer forms and particle sedimentation is enhanced by the emulsion drainage. In addition, emulsion drainage can be arrested or enhanced by the amount of particles embedded in the emulsion.
What is dispersed phase in liquid liquid extraction?
Dispersion is the breaking up of the intact phase of liquid into the droplets necessary for mass transfer.
What is the dispersed phase and dispersion medium in emulsion?
In emulsions,dispersed phase is that liquid which is less in amount and dispersion medium is the one which in large amount. As an example, oil and water can form, first, an oil-in-water emulsion, wherein the oil is the dispersed phase, and water is the dispersion medium.
What is called a dispersion phase Mcq?
it is the secondary layer in which the colloidal particles are dispersed. Explanation: since the colloidal particles require a layer in which it has to be dispersed, the dispersion phase is considered to be a second layer.
What is dispersed phase 12?
The phase that is scattered or present in the form of colloidal particles is called the dispersed phase.
What is the dispersed phase in milk class 12?
In milk, liquid fat is dispersed in water. So fat is the dispersed phase and water is the dispersion medium.
What is the dispersed phase in shaving cream?
Shaving cream is a colloid in which dispersed phase is: Gas.
What is the dispersed phase in sol?
SystemDispersed PhaseDispersion MediumSolSolidLiquidEmulsionLiquidLiquidSolid EmulsionLiquidSolidFoamGasLiquid
What are the 3 types of disperse system?
DISPERSED SYSTEMS CLASSIFIED BY THEIR PHASES In addition to the macromolecular dispersions, liquid-in-liquid emulsions, and solid-in-liquid suspensions mentioned, they include solid-in-solid suspensions (e.g., some suppositories), semisolid emulsions (e.g., creams), and gas-in-liquid foams.
What is DP and DM present in fog and clouds?
DP and DM represents dispersed phase and dispersion medium.
Which is the dispersed phase in cheese?
Dear student,cheese is liquid in solid.So,Dispersed phase is liquid and the dispersion medium is gel or solid emulsion in cheese.
What is the dispersed phase and dispersion medium of starch solution?
Example: Starch represents the dispersed phase in a starch solution, while water is the dispersing medium.
What is the dispersed phase and dispersion medium of Coloured gemstone?
(c) Coloured gem stone : Solids act as the dispersed phase as well as the dispersion medium.
Why does an emulsion separate?
Why do emulsions break? Making an emulsion is fairly easy, but it can be a little delicate. Often if the temperature is too high or the olive oil is added too quickly then the mixture can lose its ability to hold together. When this happens, the emulsification has “broken” or “separated.”
What are the two 2 stages of emulsion?
There are generally two distinct forms of two-phase emulsion: (i) water-in-oil (W/O) and (ii) oil-in-water (O/W). An emulsion that consists of water droplets dispersed in an oil phase is known as W/O emulsion, and the emulsion that consists of oil droplets dispersed in an aqueous phase is known as O/W emulsion.
What is an emulsion?
emulsion, in physical chemistry, mixture of two or more liquids in which one is present as droplets, of microscopic or ultramicroscopic size, distributed throughout the other. … Unstable emulsions eventually separate into two liquid layers.
Which is dispersed phase in emulation?
In an emulsion, one liquid (the dispersed phase) is dispersed in the other (the continuous phase). Examples of emulsions include vinaigrettes, homogenized milk, liquid biomolecular condensates, and some cutting fluids for metal working. Two liquids can form different types of emulsions.
What is extract and raffinate phase?
Liquid extraction is the separation of the constituents of a liquid by contact with another insoluble liquid called solvent. The constituents get distributed between the two phases. The solvent rich phase is called extract and the residual liquid from which the solute has been removed is called raffinate.
What is leaching in pharmacy?
From Wikipedia, the free encyclopedia. Leaching is the process of a solute becoming detached or extracted from its carrier substance by way of a solvent.