Barium sulfate is a white crystalline powder.
What is the Colour of baso4 precipitate?
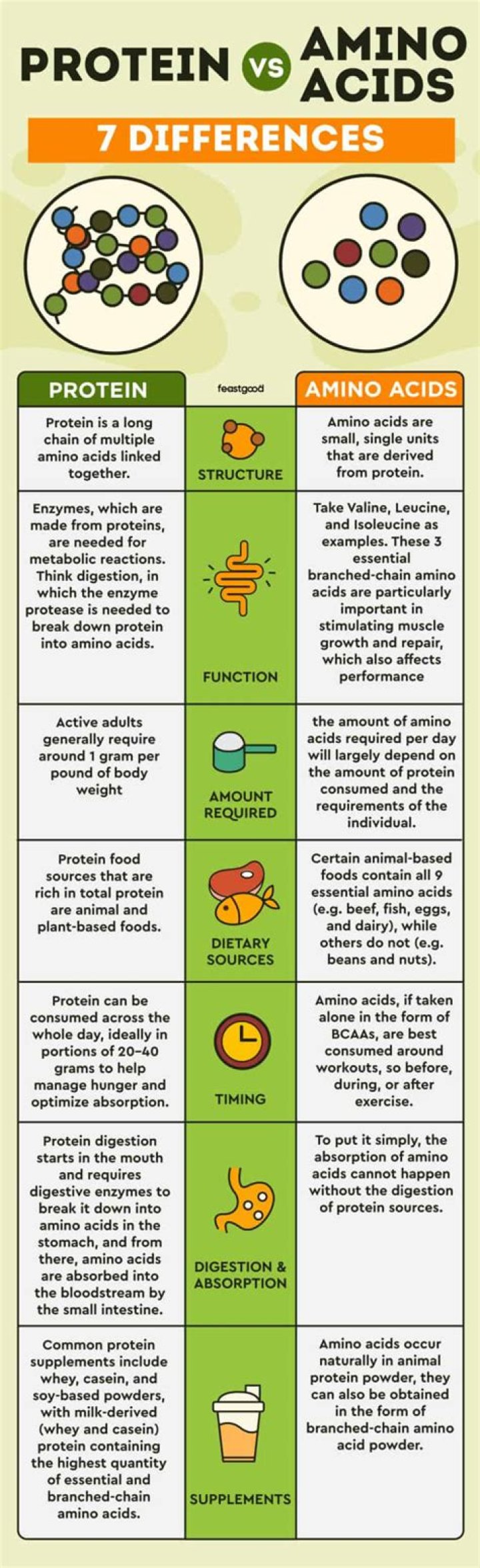
The precipitate of barium sulphate is white in colour as the salt does not release any free electrons that undergo transitions in the visible energy range.
What is the Colour of barium?
Barium is a soft, silvery-white metal, with a slight golden shade when ultrapure. The silvery-white color of barium metal rapidly vanishes upon oxidation in air yielding a dark gray layer containing the oxide.
What is the Colour of baso4 in gravimetric estimation?
On mixing a solution of barium chloride with sodium sulphate, a white precipitate of barium sulphate is immediately formed.Is baso4 a white precipitate?
Allow BaSO4 precipitate to settle (pure BaSO4 is a clean, white precipitate). Recover precipitate on 0.45um MCE (Mixed Cellulose Ester) type membranes.
What is the chemical name for baso4?
ChEBI Namebarium sulfateChEBI IDCHEBI:133326
What is the yellow precipitate?
Among the reactions given, when lead nitrate reacts with potassium iodide it gives lead nitrate and a precipitate of lead iodide is formed which is yellow in colour.
What is the Colour of sodium sulphate?
Sodium sulfate (also known as sodium sulphate or sulfate of soda) is the inorganic compound with formula Na2SO4 as well as several related hydrates. All forms are white solids that are highly soluble in water.What is the Colour of na2so4?
WHITE HYGROSCOPIC SOLID IN VARIOUS FORMS.
Which method has been used to estimate BA as baso4?DETERMINATION OF SULPHATE AS BARIUM SULPHATE USING GRAVIMETRY.
Article first time published onWhat is Colour of cucl2?
Copper chloride appears as a yellowish-brown powder (the anhydrous form) or a green crystalline solid (the dihydrate).
What is the color of calcium?
Calcium is a silvery-white, soft metal that tarnishes rapidly in air and reacts with water.
Is copper a Colour?
Copper is a reddish brown color that resembles the metal copper. The first recorded use of copper as a color name in English was in 1594.
Is BaSO4 white PPT?
where (BaSo4) or( Barium sulphate )is the white precipitate which stays back.
Is BaSO4 a salt?
BaSO4 is known as barium sulfate. This is a salt. A salt can be formed from the acids and bases reaction. If we monitor the BaSO4 development reaction, we can see that Ba^2 + comes from the base, and that SO4^2- comes from the acid.
Is BaSO4 anhydrous?
IdentifiersDensity4.49 g/cm3Melting point1,580 °C (2,880 °F; 1,850 K)Boiling point1,600 °C (2,910 °F; 1,870 K) (decomposes)
What is brown precipitate?
To tell whether an unknown substance contains iron(II) nitrate or iron(III) nitrate, add a few drops of sodium hydroxide solution: if you get a green precipitate, the unknown substance is iron(II) nitrate. if you get an orange-brown precipitate, the unknown substance is iron(III) nitrate.
What is yellow PPT?
A precipitate is a solid product that forms from a reaction and settles out of a liquid solution. The formation of a precipitate is an indication of a chemical reaction. A yellow precipitate of solid lead (II) iodide forms immediately when solutions of lead (II) nitrate and potassium iodide are mixed.
Which precipitate is white in Colour?
Metal ionResult on adding NaOH or NH 3 solutioncopper(II), Cu 2+blue precipitateiron(II), Fe 2+green precipitateiron(III), Fe 3+brown precipitatemagnesium, Mg 2+white precipitate
Is BaSO4 ionic or covalent?
Explanation: Ionic compounds are formed when a cation reacts with an anion, where electrons are “donated”. For covalent compounds, electrons are shared and only occurs between anions. BaSO4 (Barium sulfate) is an ionic compound.
What is the color of znso4?
Anhydrous zinc sulfate is a colorless crystalline solid.
What is the color of cuso4?
A copper sulphate solution is blue or bluish – green in color. Copper sulphate has also been called blue vitriol, blue – stone and roman vitriol.
What color is Naoh?
At room temperature, sodium hydroxide is a white, odorless solid. Liquid sodium hydroxide is colorless and has no odor. It can react violently with strong acids and with water.
Which Colour is iron sulphate?
The color of ferrous sulphate crystals is green. Ferrous sulphate crystals contain water molecules (FeSO4.
What Colour is sodium?
Sodium is a very soft silvery-white metal.
What is the Colour of nacl?
Pure sodium chloride is colorless, but if it contains impurities, it may take on other colors. For example, it may be purple or blue, yellow or pink.
What is gravimetry principle?
The principle behind gravimetric analysis is that the mass of an ion in a pure compound can be determined and then used to find the mass percent of the same ion in a known quantity of an impure compound. The ion being analysed is completely precipitated. The precipitate must be a pure compound.
Is BA a metal?
Barium is a chemical element with symbol Ba and atomic number 56. Classified as an alkaline earth metal, Barium is a solid at room temperature.
What is the purpose of adding HCl to the sample solution of baso4?
Barium salts can react with water to make Ba(OH)2, which is not highly soluble, and may remove some of the Ba2+ ions before they are precipitated with SO4. HCl can be added to prevent this by removing any Ba(OH)2 and changing it back to BaCl2.
What colour is CuCl4 2?
When concentrated hydrochloric acid (HCl) is added, tetrachlorocuprate II complex ions ((CuCl4)2-) are formed. These ions are yellow in colour, which with the blue colour of copper ions results in a vivid green coloured solution (centre left).
What chloride is green?
Copper(II) chloride imparts a bright green color to a flame.