A spontaneous process is capable of proceeding in a given direction without needing to be driven by an outside source of energy. … An endergonic reaction (also called a nonspontaneous reaction) is a chemical reaction in which the standard change in free energy is positive and energy is absorbed.
What is the example of non-spontaneous reaction?
Non-spontaneous Process Suppose a chemical reaction does not take place in a system. In that case, the system can still take energy and undergo a physical process. Such a process is known as a non-spontaneous process. Melting ice into water and boiling water into steam are examples of non-spontaneous processes.
What is non-spontaneous chemical reaction?
Non- Spontaneous reactions are those chemical reactions that require an energy input to proceed or that cannot take place without the influence of external factors. It requires energy input to proceed. … Examples for non-spontaneous reactions are: Diffusion of gas from low pressure to a high pressure.
What is a spontaneous reaction in chemistry?
In chemistry, a spontaneous processes is one that occurs without the addition of external energy. A spontaneous process may take place quickly or slowly, because spontaneity is not related to kinetics or reaction rate.How do I know if a reaction is spontaneous or Nonspontaneous?
If the Gibbs Free Energy is negative, then the reaction is spontaneous, and if it is positive, then it is nonspontaneous.
What is Delta G in chemistry?
In other words, ΔG is the change in free energy of a system as it goes from some initial state, such as all reactants, to some other, final state, such as all products. This value tells us the maximum usable energy released (or absorbed) in going from the initial to the final state.
Is melting of ice spontaneous?
In ice, the molecules are very well ordered because of the H-bonds. … As ice melts, the intermolecular forces are broken (requires energy), but the order is interrupted (so entropy increases). Water is more random than ice, so ice spontaneously melts at room temperature.
Which describes a non spontaneous process?
An endergonic reaction (also called a nonspontaneous reaction) is a chemical reaction in which the standard change in free energy is positive and energy is absorbed.Can non spontaneous reactions occur?
A non-spontaneous or endoergonic process is a process that can’t occur by itself, without any external driving force. … The reaction should be spontaneous, being the opposite of the non-spontaneous reaction of the previous example, but it doesn’t start.
Is a spontaneous reaction exothermic?A spontaneous reaction is more likely to be exothermic but can be endothermic. Non-spontaneous reactions are more likely to be endothermic but can be exothermic. The deciding factor for these systems is the temperature.
Article first time published onWhat is spontaneous process Class 12?
Process which takes place itself, without any external aid under the given condition is called spontaneous process.
Why are non spontaneous reactions spontaneous?
A non-spontaneous reaction can be made spontaneous when an endothermic reaction has a positive change in entropy and the temperature is greatly increased.
What is spontaneous process give example?
Definition of a Spontaneous Process A spontaneous process is one that occurs on its own, without any energy input from the outside. For example, a ball will roll down an incline; water will flow downhill; ice will melt into water; radioisotopes will decay; and iron will rust.
What is Delta G when spontaneous?
When delta G < 0 – It’s a spontaneous reaction. When delta G = 0 – It’s at equilibrium.
What is entropy and how does it relate to spontaneity of reactions?
Entropy ( S ) is a measure of the disorder in a system. … If entropy (disorder) increases, and the reaction enthalpy is exothermic ( ΔH<0 ) or weakly endothermic ( ΔH>0 & small), the reaction is generally spontaneous.
Is k1 spontaneous?
In order for lnK to be negative, K < 1. delta Go is the standard-state free energy. When this is negative, the reaction is spontaneous, therefore k is greater than one because more product is produced.
Is rusting spontaneous or Nonspontaneous?
Heat flows from a hotter object to a colder one but never reverses the process. Iron exposed to water and oxygen forms rust, but rust never spontaneously reverts to iron. A large number of exothermic reactions are spontaneous.
Is boiling spontaneous?
Why? At its boiling point liquid and vapour form are in equilibrium. Hence change in free energy is zero and the process is not spontaneous. At temperatures above the boiling point, 100oC, the process of liquid water changing to vapour is spontaneous as the process leads to increase in entropy.
What is Gibbs energy class 11?
Gibbs Energy is the maximum (or reversible) work that a thermodynamic system can perform at a constant temperature and pressure. The reversible work in thermodynamics implies a special method in which work is carried out such that the system remains in perfect equilibrium with all its surroundings.
What does ∆ s mean?
∆S is the change in entropy (disorder) from reactants to products. R is the gas constant (always positive) T is the absolute temperature (Kelvin, always positive) What it means: If ∆H is negative, this means that the reaction gives off heat from reactants to products.
What does Delta's mean?
Delta S is entropy. It’s a measurement of randomness or disorder. … We can only measure the change it undergoes through a chemical process. You cannot have this H by itself, we can’t measure it.
What is a spontaneous and non spontaneous process mention the conditions for a reaction to be spontaneous at constant temperature and pressure?
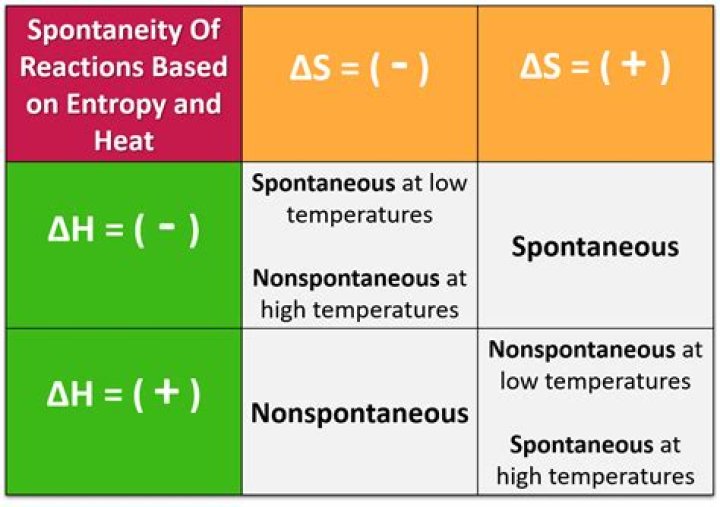
When ΔS > 0 and ΔH < 0, the process is always spontaneous as written. When ΔS < 0 and ΔH > 0, the process is never spontaneous, but the reverse process is always spontaneous. When ΔS > 0 and ΔH > 0, the process will be spontaneous at high temperatures and non-spontaneous at low temperatures.
Is Climbing stairs non spontaneous?
Climbing stairs, converting table salt to its elements, and the decomposition of water into its elements are nonspontaneous processes. Dissolving salt in water is a __process. input of energy. … Which of the following processes has a negative ∆S?
What is spontaneous equation?
ΔSsys = entropy change of the system. T = Temperature of the system. This is known as the Gibbs equation. For a spontaneous process, the total entropy change, ΔStotal is always greater than zero. ΔStotal=ΔSsys + ΔSsurr.
What is entropy class 11?
Entropy is a measure of randomness or disorder of the system. The greater the randomness, higher is the entropy. Solid state has the lowest entropy, the gaseous state has the highest entropy and the liquid state has the entropy in between the two. … The change in its value during a process, is called the entropy change.
Are waterfalls spontaneous?
A spontaneous event (or reaction) is a change that occurs under a specific set of conditions. A ball rolling down a hill, the water falling down in a waterfall, and the dispersion of the smell of a perfume when sprayed in a room (expansion of a gas), are all spontaneous events.
Is spontaneous positive or negative?
A spontaneous reaction is one that releases free energy, and so the sign of ΔG must be negative. Since both ΔH and ΔS can be either positive or negative, depending on the characteristics of the particular reaction, there are four different possible combinations.
When Delta H is positive and Delta S is negative?
When ΔH is positive and ΔS is negative, the sign of ΔG will always be positive, and the reaction can never be spontaneous. This corresponds to both driving forces working against product formation. When one driving force favors the reaction, but the other does not, it is the temperature that determines the sign of ΔG.
Is the reaction spontaneous at 25 C?
The resulting positive value of ΔG indicates that the reaction is not spontaneous at 25°C. Step 3: Think about your result. The unfavorable increase in enthalpy outweighed the favorable increase in entropy. The reaction will be spontaneous only at a more elevated temperature.
What is enthalpy and entropy?
Explanation: Enthalpy is the amount of internal energy contained in a compound whereas entropy is the amount of intrinsic disorder within the compound. … Entropy, or the amount of disorder, is always highest for gases and lowest for solids.