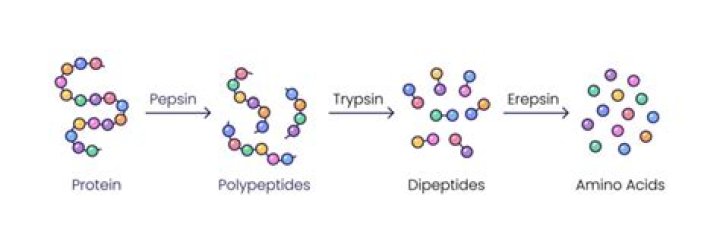
Of these five components, pepsin is the principal enzyme involved in protein digestion. It breaks down proteins into smaller peptides and amino acids that can be easily absorbed in the small intestine.
What enzyme breaks polypeptides into smaller peptides?
A protease (also called a peptidase or proteinase) is an enzyme that catalyzes (increases reaction rate or “speeds up”) proteolysis, breaking down proteins into smaller polypeptides or single amino acids, and spurring the formation of new protein products.
What enzyme breaks down polypeptide chains?
An enzyme, called pepsin, produced by cells lining the wall of the stomach, starts to attack some of the peptide bonds and splits the long protein chains into shorter polypeptides.
What enzyme converts polypeptides to peptides?
proteolytic enzyme, also called protease, proteinase, or peptidase, any of a group of enzymes that break the long chainlike molecules of proteins into shorter fragments (peptides) and eventually into their components, amino acids.What breaks down proteins into smaller polypeptide chains?
Once a protein source reaches your stomach, hydrochloric acid and enzymes called proteases break it down into smaller chains of amino acids. Amino acids are joined together by peptides, which are broken by proteases.
What type of enzyme break peptides into dipeptides and tripeptides?
The two major pancreatic enzymes that digest proteins are trypsin and chymotrypsin. Amino acids, dipeptides, and tripeptides are absorbed into the cells of the intestinal wall. The cells that line the small intestine release peptidases (enzymes) that break down dipeptides and tripeptides into single amino acids.
What enzyme breaks down polypeptides into amino acids?
Proteases are enzymes that break the peptide bond that joins amino acids together in proteins.
What is peptidase pepsin?
In context|enzyme|lang=en terms the difference between pepsin and peptidase. is that pepsin is (enzyme) a digestive enzyme that chemically digests, or breaks down, proteins into shorter chains of amino acids while peptidase is (enzyme) any enzyme that catalyzes the hydrolysis of peptides into amino acids; a protease.Which enzyme converts the protein and peptone into polypeptides and amino acid?
The enzymes that convert proteins and peptones into amino acids are the proteases.
Is protease an enzyme?Overview. Proteolytic enzymes (proteases) are enzymes that break down protein. These enzymes are made by animals, plants, fungi, and bacteria. Some proteolytic enzymes that may be found in supplements include bromelain, chymotrypsin, ficin, papain, serrapeptase, and trypsin.
Article first time published onWhere are polypeptides broken down?
Pepsin is an enzyme in the stomach that breaks down the peptide bonds in protein. In the small intestine, pancreatic enzymes that we previously learned about, called trypsin, chymotrypsin, and carboxypeptidase, really go to work breaking down the polypeptides. These enzymes enter the duodenum via the pancreatic duct.
What are peptides and polypeptides?
A peptide is a short chain of amino acids. … Peptides are generally considered to be short chains of two or more amino acids. Meanwhile, proteins are long molecules made up of multiple peptide subunits, and are also known as polypeptides. Proteins can be digested by enzymes (other proteins) into short peptide fragments.
What is the name of the enzyme that breaks down carbohydrates?
Saliva releases an enzyme called amylase, which begins the breakdown process of the sugars in the carbohydrates you’re eating.
Which enzymes break down lipids?
Lipase is an enzyme the body uses to break down fats in food so they can be absorbed in the intestines. Lipase is produced in the pancreas, mouth, and stomach.
What types of enzymes are responsible for hydrolyzing the proteins in foods?
Among these enzymes, proteases are the most important ones responsible for hydrolyzing dietary proteins and breaking them into smaller peptides and free amino acids.
What is protease degradation?
Definition. Proteolytic degradation designates the hydrolysis of one or more peptide bonds of a protein by the action of proteases. Proteolytic degradation is involved in many physiologic operations, e.g. apoptosis, cell signalling, protein maturation or turnover.
What does serine protease do?
Serine proteases are enzymes that cleave peptide bonds in proteins, in which serine serves as the nucleophilic amino acid at the active site.
Where is the protease enzyme found?
Region of digestive systemEnzymeWhere producedStomachProtease – pepsinGastric glands in stomachSmall intestine – DuodenumProtease – trypsinPancreasSmall intestine – IleumProtease – peptidaseWall of ileum
What is C3H7NO3?
Serine | C3H7NO3 – PubChem.
What does trypsin act?
Trypsin is an enzyme that helps us digest protein. In the small intestine, trypsin breaks down proteins, continuing the process of digestion that began in the stomach. It may also be referred to as a proteolytic enzyme, or proteinase.
What breaks fat into smaller pieces?
Absorption of Fats in Small Intestine: The small intestine uses bile to emulsify and break down large fat globules into smaller pieces, which allows the lipase enzymes to break down the lipids into fatty acids.
Does pepsin activate pepsinogen?
Pepsinogens are synthesized and secreted primarily by the gastric chief cells of the human stomach before being converted into the proteolytic enzyme pepsin, which is crucial for digestive processes in the stomach. … Furthermore, pepsin can activate additional pepsinogen autocatalytically.
Which enzymes convert polypeptides to Tripeptides dipeptides and amino acids?
Trypsin can activate all the proteases (including itself) and colipase (involved in fat digestion)1 as shown in the 2 figures below. The products of the action of the proteases on proteins are dipeptides, tripeptides, and individual amino acids, as shown below.
What changes protein into polypeptides?
When connected together by a series of peptide bonds, amino acids form a polypeptide, another word for protein. The polypeptide will then fold into a specific conformation depending on the interactions (dashed lines) between its amino acid side chains.
What does trypsin bind to?
Trypsin is a medium size globular protein that functions as a pancreatic serine protease. This enzyme hydrolyzes bonds by cleaving peptides on the C-terminal side of the amino acid residues lysine and arginine.
Is pepsin a protease enzyme?
Pepsin, the first animal enzyme discovered (Florkin, 1957), is an acidic protease that catalyzes the breakdown of proteins into peptides in the stomach, while it does not digest the body’s own proteins.
What does Trypsinogen breakdown?
Trypsinogen is a substance that is normally produced in the pancreas and released into the small intestine. Trypsinogen is converted to trypsin. Then it starts the process needed to break down proteins into their building blocks (called amino acids).
How does HCl activate pepsinogen?
The hormone gastrin and the vagus nerve trigger the release of both pepsinogen and HCl from the stomach lining when food is ingested. Hydrochloric acid creates an acidic environment, which allows pepsinogen to unfold and cleave itself in an autocatalytic fashion, thereby generating pepsin (the active form).
What is protease Thera blend?
Protease Thera-blend™ Breaks down protein. Bonds with alpha 2-macroglobulin to support immune function when taken on an empty stomach* Protease Thera-blend™ consists of no less than 3 strains of protease. Measured in HUT (Hemoglobin Units in a Tyrosine Base)
What does deoxyribonuclease break down?
Deoxyribonuclease (DNase) is an enzyme that breaks up extracellular DNA found in the purulent sputum during respiratory infections.
Who forms the enzymes of lysosomes?
Lysosomal enzymes are synthesized in the endoplasmic reticulum (ER), are transported to the Golgi apparatus, and are tagged for lysosomes by the addition of mannose-6-phosphate label.