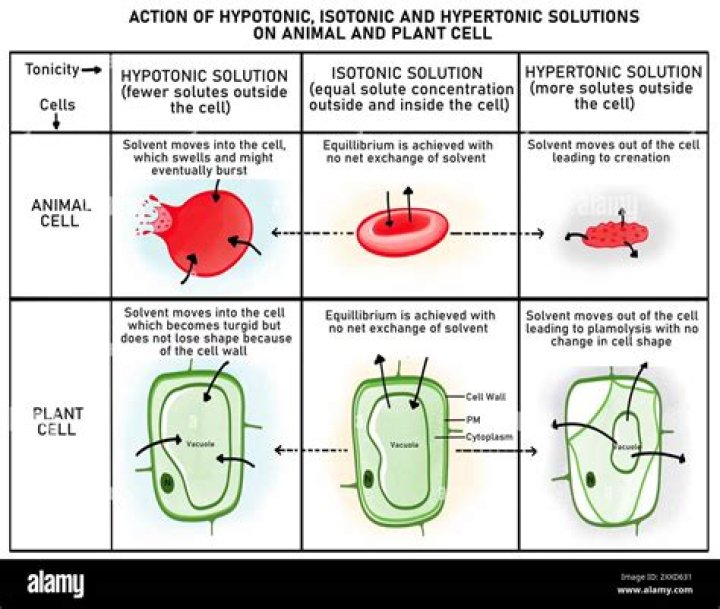
Hypotonic solutions have more water than a cell. Tapwater and pure water are hypotonic. A single animal cell ( like a red blood cell) placed in a hypotonic solution will fill up with water and then burst.
What happens when a cell is in a hypotonic solution?
A hypotonic tonic solution is any external solution that has a low solute concentration and high water concentration compared to body fluids. … A cell placed into a hypotonic solution will swell and expand until it eventually burst through a process known as cytolysis.
What happens to a cell in a hypotonic solution quizlet?
A hypotonic solution has less solute concentration and more solvent concentration. When a cell is placed in hypotonic solution , water enters the cell through osmosis. Animal cells swell and burst due to absence of cell wall. This happens when a cell shrinks inside its cell wall while the cell wall remains intact.
Why do hypotonic solutions shrink cells?
A hypertonic solution has increased solute, and a net movement of water outside causing the cell to shrink. A hypotonic solution has decreased solute concentration, and a net movement of water inside the cell, causing swelling or breakage.Why do cells swell up in hypotonic solution?
If the cell is placed in a hypotonic solution, the water potential of the surrounding medium will be higher as compared to the water potential inside the cell. Thus, water will enter into the cell through osmosis and the cell will become swollen.
What happens when a cell is placed in a hypotonic solution Class 12?
If a cell is placed in a hypotonic solution, the water enters into the cell from our side which will lead to swelling of the cell. … In this case, water moves from lower concentration to higher concentration through a membrane. The process by which water moves from the outer cell area into the cell is called osmosis.
Does hypotonic shrink the cell?
In biology, a hypotonic solution has a lower concentration of solutes outside the cell than inside the cell. If the cell is hypotonic, the solution outside the cell is hypertonic. … The cell will shrink and assume an abnormal notched shape.
What is the effect of hypotonic solution on cells with cell walls quizlet?
When the plant cell is placed in a hypotonic solution , it takes up water by osmosis and starts to swell, but the cell wall prevents it from bursting. The plant cell is said to have become “turgid” i.e. swollen and hard. The pressure inside the cell rises until this internal pressure is equal to the pressure outside.What does it mean when a cell is hypotonic to its environment?
If a cell encounters a hypotonic environment, (like pure water for instance), water will diffuse into the cell and the cell will begin to swell. … This results in a counter pressure to the pressure of the diffusing water. The amount of counter pressure needed to stop the diffusion of water is called the osmotic pressure.
How would a hypertonic solution affect a cell how could this affect cellular processes?A hypertonic solution means the environment outside of the cell has more dissolved material than inside of the cell. If a cell is placed in a hypertonic solution, water will leave the cell. This can cause a cell to shrink and shrivel.
Article first time published onWhat would happen to a prokaryotic cell in a hypotonic solution What role does the cell wall play?
As an extra, the bacterial’s cell wall helps it prevent from too much water entering in, which helps in preventing it from rupturing, but since there is penicillin inside the solution, it weakens the cell wall, and the bacteria therefore can still burst.
What will happen to the cell when the cell is placed in hypotonic solution and hypertonic solution?
If a cell is placed in a hypertonic solution, water will leave the cell, and the cell will shrink. In an isotonic environment, there is no net water movement, so there is no change in the size of the cell. When a cell is placed in a hypotonic environment, water will enter the cell, and the cell will swell.
Which cell burst up when placed in hypotonic solution?
Answer : The cells of plant, fungi and bacteria can withstand in hypotonic solution without bursting. This is because of cell wall. While animal cell bursts up in hypotonic solution because cell wall is not found.
What happens when a hypotonic solution is separated from a hypertonic solution by an osmotic membrane?
What happens when a hypotonic solution is separated from a hypertonic solution by an osmotic membrane? Water molecules move from the hypotonic solution to the hypertonic solution. A solution that is 1 ppm contains more dissolved solute than one that is 1 ppt. A typical blood serum concentration of HPO42− is 2 meq/L.
What do hypertonic solutions do?
Hypertonic solutions are ones that have a higher solute concentration than that of the cell. Hypertonic solutions cause cells to shrivel and shrink in size, which can cause problems and inhibit proper cell functioning. … Maintaining the correct balance of water and solutes will ensure that your body stays healthy.
What happens if the cell bursts?
Cytolysis, also known as osmotic lysis, occurs when a cell bursts and releases its contents into the extracellular environment due to a great influx of water into the cell, far exceeding the capacity of the cell membrane to contain the extra volume.
What will be the effect of placing a plant cell in a hypotonic solution quizlet?
What happens when a plant cell is placed in a hypotonic solution? The greatest concentration of water is outside the cell. Therefore, water enters the cell and fills the central vacuole, causing the contents of the plant cell to press against the cell wall.
Does hypotonic solution cause hemolysis?
Hypotonic solutions lead to cell swelling and eventual rupture or lysis if the resultant osmotic movement of water is great enough. In the case of red blood cells, this is referred to as hemolysis (4).
What does hypertonic and hypotonic mean?
In comparing two solutions of unequal solute concentration, the solution with the higher solute concentration is hypertonic, and the solution with the lower solute concentration is hypotonic.
What will happen to a bacterial cell if it is placed into a hypotonic environment?
When a microorganism is placed into a hypotonic environment/medium in which the concentration of solute is lower outside the cell than inside the cell, water flows into the cell. If this influx of water is uncontrolled, the cell may eventually burst.
How does hypotonic solution affect the human body?
When a hypotonic solution is administered, it puts more water in the serum than is found inside cells. As a result, water moves into the cells, causing them to swell.
Why don t bacterial cells burst in a hypotonic solution quizlet?
Why don’t bacteria lyse when placed in a hypotonic solution? Bacteria have a rigid cell wall that restricts membrane expansion and prevents lysis.
Will cheek cell burst in hypotonic solution?
Placing a cheek cell in a hypotonic solution will cause water to rush into the cell, which causes the cell to burst open and release its DNA.
In what solution does the cell swell up?
In the hypotonic solution, the cell will gain water by osmosis and will swell up.
Which statement characterizes a hypotonic solution?
The solution in which the concentration of solutes is less and concentration of solvent (water) is less as compared to the specific cell.
What happens when two solutions separate?
What happens when two solutions separated by a selectively permeable membrane reach osmotic equilibrium? Water molecules move between the two solutions, but there is no net movement of water across the membrane. Water molecules are constantly in motion and will continually move across the membrane.
What causes osmosis?
Osmosis occurs according to the concentration gradient of water across the membrane, which is inversely proportional to the concentration of solutes. … Osmosis occurs when there is a concentration gradient of a solute within a solution, but the membrane does not allow diffusion of the solute.