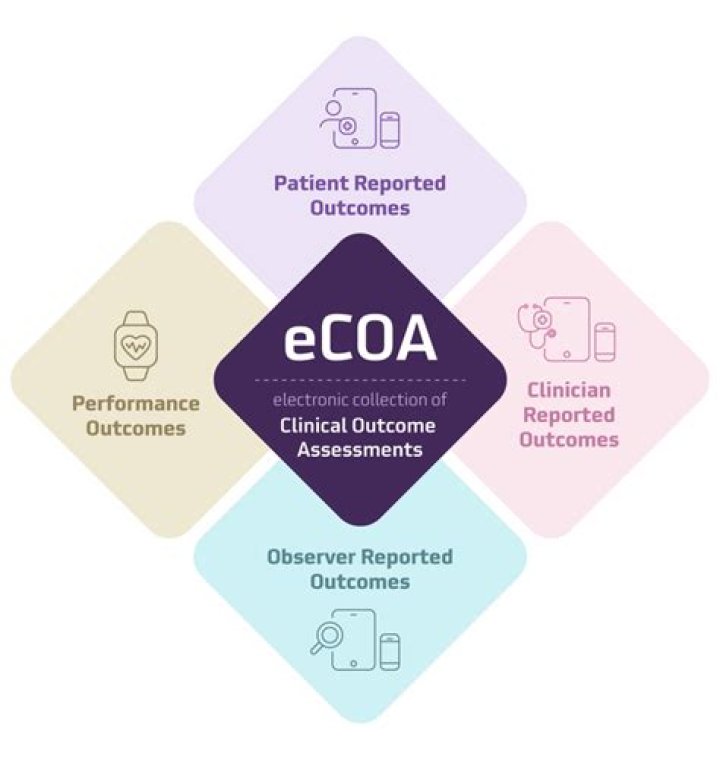
The IQVIA electronic Clinical Outcome Assessment (eCOA) solution is an advanced study build and execution platform that optimizes real-time, direct-from-patient data collection.
What is the difference between EDC and eCOA?
While DDC may seem like standard electronic data capture (EDC), they are different. … Similarly, eCOA questionnaires capture data directly from clinicians, replacing an assortment of specific forms created for a variety of research purposes that would also need to be transcribed.
What are clinician reported outcome measures?
A ClinRO is an appropriate measure to use when: A clinician can make accurate assessments of observations that reflect patient feeling, function, or predict survival. For example, a clinician listens to wheezing during a lung exam. Patients don’t typically listen to their own lungs.
What is eCOA Medidata?
Medidata eCOA (Clinical Outcome Assessment) is revolutionizing the way sponsors, CROs, and sites collect electronic data from patients, physicians and caregivers.What is clinical Rtsm?
An RTSM, or Randomization and Trial Supply Management system, is responsible for enabling critical functions of a clinical trial, from randomizing patients (who gets the active drug vs.
What is the difference between CRF and EDC?
A CRF is the standardized form on which the Clinical site (Lay People can think doctor’s office) transcribes relevant data from the medical chart (also called the source) of a clinical trial subject. … Now “True eDC” deals with data being recorded as source in an electronic format.
What is Iwrs in clinical trials?
Interactive Web Response Systems (IWRS) and Interactive Voice Response Systems (IVRS) are the technologies that research sites use to enroll patients into clinical trials, randomize patients, and manage study drug supplies.
What is eCRF research?
An eCRF (electronic case report form) is a software system used to collect data in a clinical study. Commonly, eCRFs are web-based applications containing various data forms and fields designed to receive data in clinical trials or observational studies.What is the purpose of Reg B?
Regulation B prohibits creditors from requesting and collecting specific personal information about an applicant that has no bearing on the applicant’s ability or willingness to repay the credit requested and could be used to discriminate against the applicant.
How are clinical outcomes measured?Clinical outcomes can be measured by activity data such as hospital re-admission rates, or by agreed scales and other forms of measurement. They can be recorded by administrators or by clinical staff such as doctors, nurses, psychologists or allied health professionals (e.g. physiotherapists, dietitians).
Article first time published onWhat are examples of outcome measures?
Outcome measures reflect the impact of the health care service or intervention on the health status of patients. For example: The percentage of patients who died as a result of surgery (surgical mortality rates). The rate of surgical complications or hospital-acquired infections.
What is an outcome measurement tool?
An outcome measure is a tool used to assess a patient’s current status. Outcome measures may provide a score, an interpretation of results and at times a risk categorization of the patient. … The initial results may help determine the course of treatment intervention.
Is IRT the same as Rtsm?
Most of the terms are just different flavors of the same thing. Wouldn’t it be nice if the industry could come together and agree on a uniform term? We’ve been asked why we use agile RTSM (Randomization and Trial Supply Management) instead of IRT (Interactive Response Technology) when we talk about our software.
What is randomization and trial supply management?
Randomization & Trial Supply Management is the umbrella term for the various technologies that allow a study team to enroll patients, randomize participants, and manage drug supply ordering. … IVRS and IWRS are both used to randomize patients and manage drug supplies.
What does 4G clinical do?
4G Clinical creates software to run the back-end of a clinical trial. That means randomizing patients into treatment and placebo groups, locating medicines and placebos, keeping the supply of those medicines stocked, and, at least during the pandemic, delivering those medicines to patients.
What are EDC Systems?
What is an EDC system? To put it simply, an Electronic Data Capture (EDC) system is software that stores patient data collected in clinical trials. … Sponsors, contract research organizations (CROs), and sites have adopted EDC systems to carry out both simple and complex trials in all phases of research.
What is Medidata Rave?
Medidata Rave® is a cloud–based clinical data management system used to electronically capture, manage, and report clinical research data. … Through its iMedidata® portal, users have fast, simple access to all studies, Medidata applications, eLearning training courses, online discussions, and more in one place.
What is an IVR line?
Interactive Voice Response (IVR) is an automated phone system technology that allows incoming callers to access information via a voice response system of pre recorded messages without having to speak to an agent, as well as to utilize menu options via touch tone keypad selection or speech recognition to have their …
What is vendor in clinical trial?
A travel vendor can ensure the patient attends all study appointments by providing services like ground transportation or medical-assisted transportation. … Some clinical trial recruitment companies work directly with travel vendors, so be sure to ask about their relationships as you line up your vendor team.
What is OC RDC?
Oracle Clinical Remote Data Capture (OC RDC)
What is InForm in clinical trials?
A data capture and study management application that uses a secure web browser to capture clinical study data and manage the clinical study process. Use InForm to screen and enroll subjects, track visits, raise and resolve queries, and evaluate clinical study data.
Does ECOA apply to deposit accounts?
Answer: Regulation B applies to credit products – so the answer would be no, as neither the regulation nor the commentary addresses the application of adverse action to non-credit products. However, the amended Section 615(a) of the FCRA would apply to any type of adverse action (deposit accounts, insurance, etc.)
Who is subject to the ECOA and regulation B requirements?
The Equal Credit Opportunity Act (ECOA), 15 U.S.C. § 1691 et seq. , which is implemented by Regulation B (12 CFR Part 1002 ), applies to all creditors, including credit unions. When originally enacted, ECOA gave the Federal Reserve Board responsibility for prescribing the implementing regulation.
Does ECOA apply to Helocs?
Both the ECOA and the FCRA have adverse action requirements that may apply when a creditor suspends a HELOC or reduces the credit limit because of a significant decline in the value of a property.
How many Cros are there in the world?
There are more than 1,100 CRO companies around the globe, led by IQVIA, Covance, LabCorp, Syneos, Charles River, Parexel, ICON, PRA, PPD and Wuxi AppTec. The world’s top ten CRO players hold a combined over 50% market share for now.
What is a casebook in clinical trials?
Casebook: A detailed report of the diagnosis, treatment, and follow-up of an individual patient. Case reports also contain some demographic information about the patient (for example, age, gender, ethnic origin..). Event: Visit (See definition in Clinical Trial Terminology)
What is TMF Reference Model?
The Trial Master File (TMF) Reference Model is a supported initiative of the Drug Information Association’s (DIA) Document and Records Management Community and represents a single, unified interpretation of the regulations and best practices related to Trial Master Files that would be accepted by all clinical trial …
What are the four measures of health?
- Physical, mental, and social health–related quality of life.
- Well-being/satisfaction.
- Participation in common activities.
What are examples of clinical outcomes?
Typical examples of outcomes are cure, clinical worsening, and mortality. The primary outcome is the variable that is the most relevant to answer the research question. Ideally, it should be patient-centered (i.e., an outcome that matters to patients, such as quality of life and survival).
What are metrics in healthcare?
A healthcare KPI or metric is a well-defined performance measurement that is used to monitor, analyze and optimize all relevant healthcare processes to increase patient satisfaction. Many of these metrics are actually specific key performance indicators for hospitals.
What is the difference between process and results measures?
Process measures can determine the volume at which cost-effective preventive services are performed, while outcome measures give a payer insight into the relationship between preventive services and healthier outcomes.