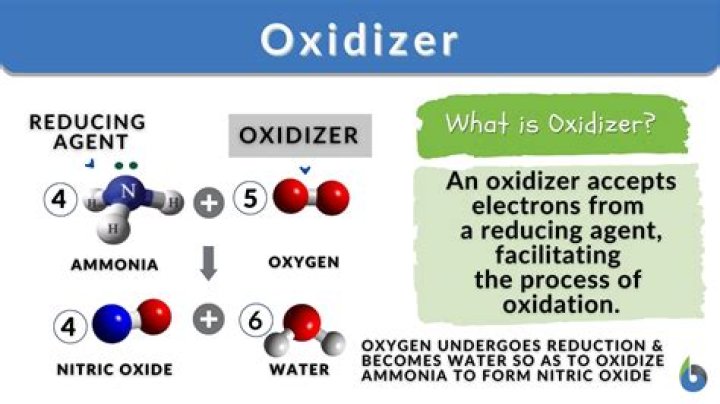
An oxidizing agent, also known as an oxidant or oxidizer, is a substance that has the ability to oxidize other substances — in other words to accept their electrons. Common oxidizing agents are oxygen, hydrogen peroxide and the halogens.
What is the meaning of oxidising in chemistry?
verb. to undergo or cause to undergo a chemical reaction with oxygen, as in formation of an oxide. to form or cause to form a layer of metal oxide, as in rusting. to lose or cause to lose hydrogen atoms.
What is oxidation class 10th?
Oxidation refers to the loss of electrons or increase in oxidation state by a molecule, atom, or ion. Reduction refers to the gain of electrons or decrease in oxidation state by a molecule, atom, or ion.
Why is it called oxidation?
Oxidation means the process of losing of electrons by any substance. The substance which loses election in the process is called reductant or oxidising agent. Oxidation also means the process of addition of oxygen. Since oxygen is being added to the compound, so the process is called oxidation.What is oxidation Class 11?
Oxidation is defined as the addition of oxygen/electronegative element to a substance or rememoval of hydrogen/ electropositive element from a susbtance.
What is oxidation in geography?
Oxidation – the breakdown of rock by oxygen and water, often giving iron-rich rocks a rusty-coloured weathered surface.
What is oxidation and example?
The term oxidation was originally used to describe reactions in which an element combines with oxygen. Example: The reaction between magnesium metal and oxygen to form magnesium oxide involves the oxidation of magnesium. … In the course of this reaction, each magnesium atom loses two electrons to form an Mg2+ ion.
What is oxidation in BYJU's?
When a reactant loses electrons during a reaction, it is called oxidation.What is called loss of electron?
The loss of electrons is called oxidation. The gain of electrons is called reduction. … As such, electron-transfer reactions are also called oxidation-reduction reactions, or simply redox reactions. The atom that loses electrons is oxidized, and the atom that gains electrons is reduced.
What is oxidising agent class 10 examples?Examples of oxidizing agents include halogens, potassium nitrate, and nitric acid. A reducing agent, or reductant, loses electrons and is oxidized in a chemical reaction.
Article first time published onWhat is oxidation in biology?
Biological oxidation is the combination of oxidation-reduction transformations of substances in living organisms. Oxidation-reduction reactions are those which take place with a change in the oxidation state of atoms through the redistribution of electrons between them.
What is oxidation and reduction class 12?
Oxidation reaction: The reaction in which oxygen is gained or hydrogen is lost, is called oxidation reaction. e.g. 2Cu+O2heat 2CuO. Reduction reaction. The reaction in which hydrogen is gained or oxygen is lost, is called reduction reaction.
What is oxidation in weathering?
Oxidation is another kind of chemical weathering that occurs when oxygen combines with another substance and creates compounds called oxides. … When rocks, particularly those with iron in them, are exposed to air and water, the iron undergoes oxidation, which can weaken the rocks and make them crumble.
What is known as oxidation Class 9?
Answer. Oxidation means reaction of minerals in rocks to atmospheric oxygen. When the atmospheric oxygen comes in contact with iron-compounds in rocks, the iron starts rusting or crumbling. The colour of the rock changes to red, yellow or brown. 20 Likes.
What is known as oxidation class 9 geography?
What is known as oxidation ? Answer: It is the process of reaction of minerals present in rocks to atmospheric oxygen.
What is carbonation in GEO?
Carbonation. Carbonation is the process in which atmospheric carbon dioxide leads to solution weathering. Carbonation occurs on rocks which contain calcium carbonate such as limestone and chalk.
Where does oxidation occur?
If a chemical species loses one or more electrons, this is called oxidation. The opposite process, the gain of electrons, is called reduction. Oxidation occurs at the Anode. Reduction occurs at the Cathode.
What is removal of hydrogen?
The removal of hydrogen from a substance is called oxidation.
What is redox equation?
An oxidation-reduction (redox) reaction is a type of chemical reaction that involves a transfer of electrons between two species. … The oxidation state of an individual atom is 0. The total oxidation state of all atoms in: a neutral species is 0 and in an ion is equal to the ion charge.
Is respiration a redox reaction?
Cellular respiration is a redox reaction, which is an oxidation-reduction reaction. Respiration is a collection of metabolic reactions in which electrons are lost and gained. As a result, it’s referred to as the oxidation-reduction or redox reaction.
What is oxidation number Class 12?
The number allotted to an element in a compound representing the number of electrons lost or gained by an atom of the element of the compound is called oxidation state.
What is oxidation class 7th?
Answer: Oxidation is a chemical reaction that involves addition of oxygen or removal of hydrogen from a substance. For example, when carbon reacts with oxygen to form carbon dioxide, it is said to be oxidised by gaining oxygen.
What is an oxidizing agent answers?
An oxidizing agent (often referred to as an oxidizer or an oxidant) is a chemical species that tends to oxidize other substances, i.e. cause an increase in the oxidation state of the substance by making it lose electrons.
What is reduction answer?
Reduction involves a half-reaction in which a chemical species decreases its oxidation number, usually by gaining electrons. ★ Together, reduction and oxidation form redox reactions. (Reduction – Oxidation = REDOX)
What do you mean by oxidation and reduction?
The terms oxidation and reduction can be defined in terms of the adding or removing oxygen to a compound. … Oxidation is the gain of oxygen. Reduction is the loss of oxygen.
What is oxidation and reduction class 11?
Oxidation: Oxidation is defined as “addition of oxygen or any electronegative element and removal of hydrogen or any electropositive element”. Reduction: Reduction is defined as” addition of hydrogen or any electropositive element and removal of oxygen or any electronegative element”.