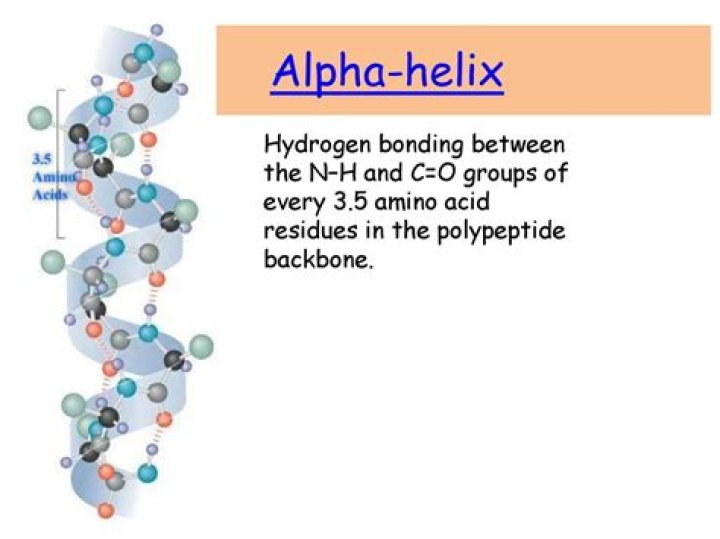
An α-helix is a right-handed coil of amino-acid residues on a polypeptide chain, typically ranging between 4 and 40 residues. … Amino acids whose R-groups are too large (tryptophan, tyrosine) or too small (glycine) destabilize α-helices.
What makes an alpha helix?
An alpha helix is a common shape that amino acid chains will form. The alpha helix is characterized by a tight right-handed twist in the amino acid chain that causes it to form a rod shape. Hydrogen bonds between the hydrogen in an amino group and the oxygen in a carboxyl group on the amino acid cause this structure.
How do you identify an alpha helix?
In an alpha helix, the main chain arranges in a right-handed helix with the side chains pointing away from the helical axis. (Stereo: ON OFF) In the following, the side chains are truncated at the beta carbon (green) to allow a better view of the main chain.
What amino acids disrupt an alpha helix structure?
Amino acids with too large R group (tryptophan, tyrosine) or too small R group (glycine) amino acids tend to disrupt alpha helices. Proline also destabilises the alpha helix and the reason is it’s irregular geometry, its R-group bonds back to the nitrogen of the amide group, which causes steric hindrance.What stabilizes an alpha helix?
1 α-Helix. An α-helix secondary structure is stabilized by hydrogen bonds between carbonyl oxygen and the amino group of every third residue in the helical turn with each helical turn consisting of 3.6 amino acid residues (Fig. 10.1A). The side chain of amino acids is projected outward from the outer helical surface.
Which amino acids make alpha helix?
Methionine, alanine, leucine, glutamate, and lysine have special propensity to be part of α-helix structures while proline and glycine have poor helix-forming propensities.
Which structure of protein is alpha helix?
The alpha helix (α-helix) is a common motif in the secondary structure of proteins and is a right hand-helix conformation in which every backbone N−H group hydrogen bonds to the backbone C=O group of the amino acid located four residues earlier along the protein sequence.
Which amino acid is alpha helix Terminator?
Glycine also occurs in beta-strands and alpha-helices, although its frequency in alpha helices is low (second lowest, after proline, p. 125 in). Glycine is commonly found at the C-terminus of alpha helices, and is considered a helix terminator (p.Which amino acids would destabilize alpha helices?
All the amino acids are found in α-helices, but glycine and proline are uncommon, as they destabilize the α-helix. Glycine is exempt from many steric constraints because it lacks a β carbon.
Which of the following amino acid is a alpha helix Terminator?Gly and Asn residues are the most frequent alpha L helix terminators, with the former having a very high propensity to adopt such conformations.
Article first time published onWhy alpha helix is right-handed?
Despite the fact that, based on the Ramachandran plot, both right-handed and left-handed alpha helices are among the permitted conformations, the right-handed alpha helix is energetically more favorable because of fewer steric clashes between the side chains and the main chain.
What is alpha helix and beta sheet?
The most common types of secondary structures are the α helix and the β pleated sheet. Both structures are held in shape by hydrogen bonds, which form between the carbonyl O of one amino acid and the amino H of another. Images showing hydrogen bonding patterns in beta pleated sheets and alpha helices.
What are turns and loops?
Loops and turns connect α helices and β strands. The most common types cause a change in direction of the polypeptide chain allowing it to fold back on itself to create a more compact structure. … Loops that have only 4 or 5 amino acid residues are called turns when they have internal hydrogen bonds.
What interactions stabilize folding of an alpha-helix?
Two major factors stabilize the alpha helix: intrachain H-bonding and minimization of steric interference between side chains.
What affects the stability of alpha-helix?
Amino acids whose R-groups are too large (tryptophan, tyrosine) or too small (glycine) destabilize α-helices. … Another factor affecting α-helix stability is the total dipole moment of the entire helix due to individual dipoles of the C=O. groups involved in hydrogen bonding.
What type of bonding provides stability to alpha-helix structure of proteins?
Therefore, Hydrogen bonding is the intermolecular bonding which helps us in stabilizing the alpha-helix structure of proteins. And H bonds are formed between -NH- of amino acid and C = O of the adjacent turn of helix.
Is Alpha Helix a tertiary structure?
For example, the α-helices may be oriented parallel to each other or at right angles. So the tertiary structure refers to the folding of the different segments of helices, sheets, turns, and the remainder of the protein into its native three-dimensional structure.
Is Alpha Helix a secondary structure?
The α-helix is a common element of protein secondary structure, formed when amino acids “wind up” to form a right-handed helix where the side-chains point out from the central coil (Fig.
What does the alpha helix protein do in a cell membrane?
α-helical membrane proteins are responsible for interactions between most cells and their environment. [5] Trans-membrane (TM) helices are typically encoded by stretches of 17-25 residues [6], which provide sufficient length to cross the membrane.
Which of the following amino acid will be absent in alpha-helix structure of protein?
Ans. ( Proline is an imino acid. The amino group of proline is covalently connected to the side chain (R group) and thus it reduces the rotational flexibility. Furthermore proline produces a kink in the polypeptide. Due to these reasons proline is very rarely present in α-helixes.
Which of the following amino acid is most compatible with an alpha helical structure?
Alanine is very favorable for formation of alpha helical structure because of amino acids with simple side chains.
Can D amino acids form alpha-helix?
The incorporation of an adjacent pair of d-amino acids into an α-helical model peptide enhances the effect of helix disturbance.
Which amino acids are typically not found in alpha helices or beta sheets and why?
Explanation: Beta bends are part of secondary protein structures. They serve as a link between alpha helices and beta sheets. Beta bends are composed of proline and glycine, amino acids that usually are not found in alpha helices.
How are alpha helices held together?
The alpha-helix is a right-handed helical coil that is held together by hydrogen bonding between every fourth amino acid.
Which protein stage contains alpha helix and beta sheets?
Secondary structure is local interactions between stretches of a polypeptide chain and includes α-helix and β-pleated sheet structures. Tertiary structure is the overall the three-dimension folding driven largely by interactions between R groups.
Which peptide would be most likely to form an alpha helix?
Peptide c is most likely to form an alpha helix with its three charged residues (Lys, Glu, and Arg) aligned on one face of the helix.
Which is 21st amino acid?
Selenocysteine: the 21st amino acid.
Which of the following is an example of Sulphur containing amino acid?
Methionine, cysteine, homocysteine, and taurine are the 4 common sulfur-containing amino acids, but only the first 2 are incorporated into proteins.
Which of the following is an amino acid?
The essential amino acids are histidine, isoleucine, leucine, lysine, methionine, phenylalanine, threonine, tryptophan, and valine. The nonessential amino acids are alanine, asparagine, aspartic acid, glutamic acid, and serine.
What holds collagen together?
Hydrogen bonding helps collagen to form its ‘tertiary’ structure. The hydrogen bonds are very strong and keep the connective tissues in the body together.
Are there left-handed alpha helix?
Proteins typically consist of right-handed alpha helices, whereas left-handed alpha helices are rare in nature. Peptides of 20 amino acids or less corresponding to protein helices do not form thermodynamically stable alpha helices in water away from protein environments.