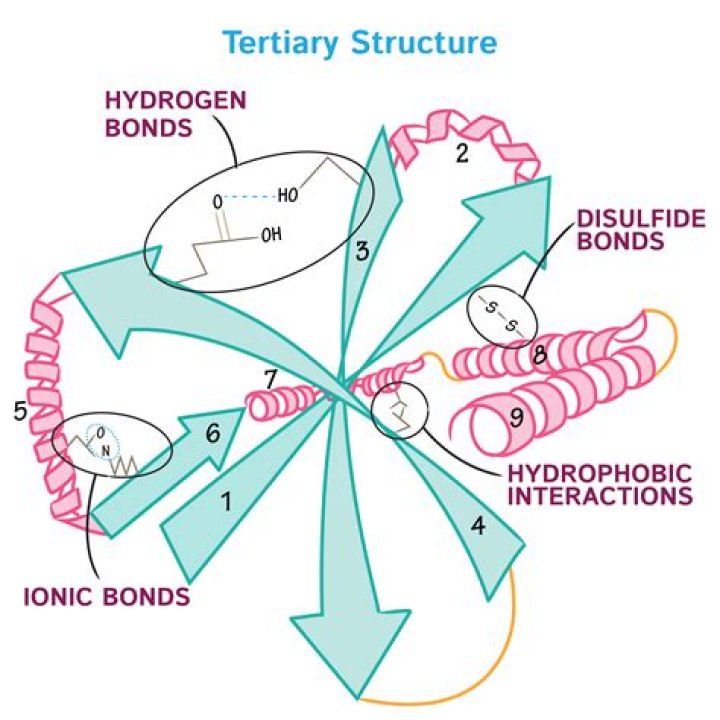
Tertiary structure is stabilized by multiple interactions, specifically side chain functional groups which involve hydrogen bonds, salt bridges, covalent disulfide bonds, and hydrophobic interactions.
What bonds are in tertiary protein structure?
The tertiary structure of a protein consists of the way a polypeptide is formed of a complex molecular shape. This is caused by R-group interactions such as ionic and hydrogen bonds, disulphide bridges, and hydrophobic & hydrophilic interactions.
What bonds are in the quaternary structure?
The quaternary structure of a protein is the association of several protein chains or subunits into a closely packed arrangement. Each of the subunits has its own primary, secondary, and tertiary structure. The subunits are held together by hydrogen bonds and van der Waals forces between nonpolar side chains.
How is the tertiary structure held together?
The tertiary structure of a protein is held together by interactions between the the side chains – the “R” groups. There are several ways this can happen. Some amino acids (such as aspartic acid and glutamic acid) contain an extra -COOH group. Some amino acids (such as lysine) contain an extra -NH2 group.Which bond is not present in tertiary protein structure?
The amino acids present in proteins differ in all of the options: size, shape, and side groups. Which of the following bonds are not involved in tertiary structure? Answer a. Peptide bonds are not involved in tertiary structure.
What bonds hold primary structures of a protein together?
Primary Structure The structure is held together by peptide bonds between the carboxyl and amino group of each amino acid, which is formed during protein translation. Primary structure is heavily dependent upon the DNA sequence within the gene that codes for that particular protein.
Are there covalent bonds in tertiary structure?
Finally, there’s one special type of covalent bond that can contribute to tertiary structure: the disulfide bond. Disulfide bonds, covalent linkages between the sulfur-containing side chains of cysteines, are much stronger than the other types of bonds that contribute to tertiary structure.
What is an example of a tertiary protein structure?
Protein tertiary structure. For example, amide hydrogen atoms can form H‐bonds with nearby carbonyl oxygens; an alpha helix or beta sheet can zip up, prompted by these small local structures. Hydrophobic interactions among the amino acid side chains also determine tertiary structure.What interactions occur in the quaternary structure?
Quaternary structure describes how polypeptide chains fit together to form a complete protein. Quaternary protein structure is held together by hydrophobic interactions, and disulfide bridges.
What type of bonds occur in tertiary and quaternary structures to cause a globular look?Hydrogen bonds between back bone atoms are important in maintaining secondary structures, and those between side chains are involved in maintaining the tertiary structure. Examples of finding and visualizing both types in globular proteins are at hydrogen bonds.
Article first time published onWhat type of bonds mostly hold the protein in its tertiary structure we will explore the importance of this later?
The tertiary structure of a protein molecule encompasses the overall folding of polypeptide chains, where, if more than one chain is present, the chains are linked by covalent bonds—most often disulfide bonds (as in II).
Which of the following are bonds that are formed during tertiary structure formation in protein folding?
Tertiary structureThe tertiary structure of proteins is determined by hydrophobic interactions, ionic bonding, hydrogen bonding, and disulfide linkages.
Why are hydrogen bonds important in tertiary structure?
Hydrogen bonds form between the oxygen of each C=O. bond in the strand and the hydrogen of each N-H group four amino acids below it in the helix. The hydrogen bonds make this structure especially stable. The side-chain substituents of the amino acids fit in beside the N-H groups.
What type of bond in the tertiary structure is the strongest?
If the two electron clouds of adjacent atoms are too close, repulsive forces come into play because of the negatively-charged electrons. Covalent bonds are the strongest chemical bonds contributing to protein structure.
What type of bonds hold the enzyme in its folded Tertiary shape?
Tertiary Structure Cysteine side chains form disulfide linkages in the presence of oxygen, the only covalent bond forming during protein folding. All of these interactions, weak and strong, determine the final three-dimensional shape of the protein.
Are peptide bonds covalent?
Examples of important covalent bonds are peptide (amide) and disulfide bonds between amino acids, and C–C, C–O, and C–N bonds within amino acids. Coordinate covalent bonds involve the unequal sharing of an electron pair by two atoms, with both electrons (originally) coming from the same atom.
How are ionic bonds involved with the tertiary structure of proteins?
Tertiary structure comprises four types of covalent and non-covalent interactions: – Hydrogen bonds of polar amino acid residues. – Ionic bonds between amino acids with oppositely charged side chains. – Hydrophobic interactions in which non-polar amino acids cluster.
What types of bonds are in primary structure?
Explanation: Primary structure consists of amino acids joined by peptide bonds. Peptide bonds are between the alpha-carboxyl of one amino acid, and the alpha-amine of the next amino acid. A peptide bond is an example of an amide bond.
Which interaction helps stabilize the tertiary level of protein organization?
The tertiary structure of a protein refers to the overall three-dimensional arrangement of its polypeptide chain in space. It is generally stabilized by outside polar hydrophilic hydrogen and ionic bond interactions, and internal hydrophobic interactions between nonpolar amino acid side chains (Fig. 4-7).
Are all enzymes quaternary?
Explanation: It would completely depend on the enzyme we’re considering. Some are only monomeric (e.g. trypsin), some contain several subunits which interact to form a quaternary structure. … In the case of trypsin, a serine protease, we could say the enzyme only has a tertiary structure.
What is an example of the quaternary structure of a protein?
The quaternary structure refers to the number and arrangement of the protein subunits with respect to one another. Examples of proteins with quaternary structure include hemoglobin, DNA polymerase, ribosomes, antibodies, and ion channels.
What causes the quaternary structure of a protein?
This structure is caused by chemical interactions between various amino acids and regions of the polypeptide. … In nature, some proteins are formed from several polypeptides, also known as subunits, and the interaction of these subunits forms the quaternary structure.
What are the main types of tertiary structure?
There are four types of tertiary interactions: hydrophobic interactions, hydrogen bonds, salt bridges, and sulfur-sulfur covalent bonds.
Are disulfide bonds covalent?
14.4. Disulfide bonds are another kind of covalent interactions that can be formed by oxidation to build up a network. They play an important role in the heat-induced gelation of globular proteins.
Where are tertiary proteins found?
Tertiary Structure: Water-Soluble Proteins Water soluble proteins, which include enzymes and transport proteins, are found free in cellular compartments such as the cytoplasm, nucleus, or endoplasmic reticulum.
What type of bonds cause a globular look?
Quaternary Structure Weak interactions between the subunits help to stabilize the overall structure. For example, insulin (a globular protein) has a combination of hydrogen bonds and disulfide bonds that cause it to be mostly clumped into a ball shape.
Is globular protein tertiary structure?
All globular proteins are water-soluble, with their tertiary level of structure often resembling a globe or sphere. This ‘globular’ folded shape is crucial to the function of these proteins and their ability to serve a variety of biological roles.
Do globular proteins have disulfide bonds?
3 Disulfide Bonds. Most globular proteins contain internal covalent cross-links in the form of disulfide bonds (cystine residues). There is relatively free rotation about the disulfide bond in simple compounds, such as dimethyl disulfide.
What are the five types of intermolecular forces that lead to tertiary structure?
- Intramolecular forces. Amino acids, monomers of proteins, form peptide bonds [red color] upload.wikimedia.org. …
- Intermolecular forces. slideplayer.com. Hydrogen bonding, ionic bonding, dipole-dipole interactions, London dispersion forces and disulfide bonds.
What holds a protein in its tertiary structure quizlet?
The tertiary structure is held together by bonds between the R groups of the amino acids in the protein, and so depends on what the sequence of amino acids is. There are three kinds of bonds involved: hydrogen bonds, which are weak.
What determines the tertiary structure of a protein quizlet?
Tertiary structure is influenced by ionic bonds between opposite charged R-groups, hydrogen bonds between R-groups bearing opposite partial charges, and hydrophobic interactions resulting from the tendency of nonpolar R-groups to stay close together in an aqueous solution.