Examples of chemical properties include flammability, toxicity, acidity, reactivity (many types), and heat of combustion. Iron, for example, combines with oxygen in the presence of water to form rust; chromium does not oxidize (Figure 2).
What are the 5 chemical properties?
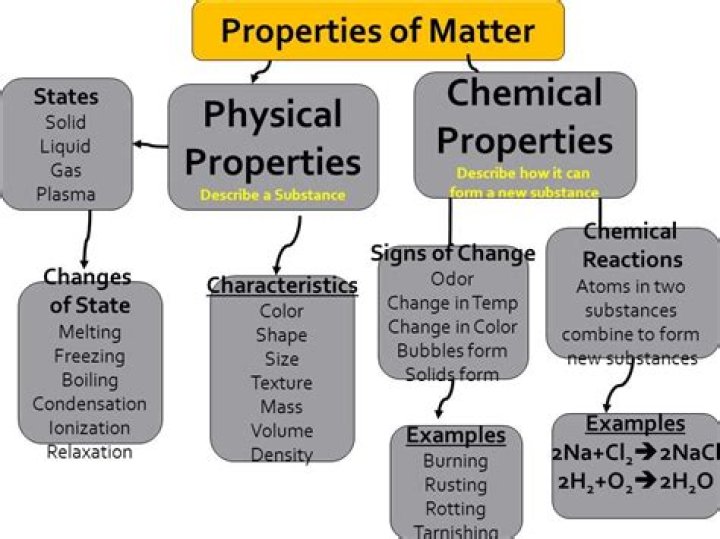
A chemical property is a characteristic of a particular substance that can be observed in a chemical reaction. Some major chemical properties include flammability, toxicity, heat of combustion, pH value, rate of radioactive decay, and chemical stability.
What are the chemical properties of matter?
Chemical properties are properties that can be measured or observed only when matter undergoes a change to become an entirely different kind of matter. They include reactivity, flammability, and the ability to rust. Reactivity is the ability of matter to react chemically with other substances.
What are 5 chemical changes matter?
Yes; new substances formed, as evidenced by the color changes and bubbles. Some signs of a chemical change are a change in color and the formation of bubbles. The five conditions of chemical change: color change, formation of a precipitate, formation of a gas, odor change, temperature change.What are 4 types of chemical properties?
Key Takeaways: Chemical Property Examples of chemical properties include flammability, toxicity, chemical stability, and heat of combustion.
What are the 13 properties of matter?
- color (intensive)
- density (intensive)
- volume (extensive)
- mass (extensive)
- boiling point (intensive): the temperature at which a substance boils.
- melting point (intensive): the temperature at which a substance melts.
What are 5 chemical properties of chlorine?
Properties: Chlorine has a melting point of -100.98°C, boiling point of -34.6°C, density of 3.214 g/l, specific gravity of 1.56 (-33.6°C), with a valence of 1, 3, 5, or 7. Chlorine is a member of the halogen group of elements and directly combines with almost all of the other elements.
What are 10 examples chemical changes?
Examples of chemical changes are burning, cooking, rusting, and rotting. Examples of physical changes are boiling, melting, freezing, and shredding. Many physical changes are reversible, if sufficient energy is supplied.What are 5 chemical changes examples?
- Burning of paper and log of wood.
- Digestion of food.
- Boiling an egg.
- Chemical battery usage.
- Electroplating a metal.
- Baking a cake.
- Milk going sour.
- Various metabolic reactions that take place in the cells.
- Combustion.
- Oxidation (rusting)
- Decomposition or fermentation.
- Cooking an egg.
- Photosynthesis.
- Reacting acids and bases together.
- Chemical batteries.
- Digestion.
What are the 7 properties of matter?
- Volume. Definition.
- Boiling point. Definition.
- Odor. Definition.
- Melting point. Definition.
- Color. Definition.
- Density. Definition.
- Texture. Definition.
What are 4 chemical properties of oxygen?
- It is a colorless, odorless and tasteless gas.
- It readily dissolves in cold water.
- It is highly reactive and form oxides with almost all elements except noble gases.
- Liquid oxygen is strongly paramagnetic.
- It exists in three allotropic forms- monoatomic, diatomic and triatomic.
What are the 3 properties of matter?
The three basic properties of matter are volume, mass, and shape. 2. All matter is made up of tiny particles called atoms. 3.
What are 5 metallic properties?
Metals are lustrous, malleable, ductile, good conductors of heat and electricity.
What are the 8 chemical properties of minerals?
Properties that help geologists identify a mineral in a rock are: color, hardness, luster, crystal forms, density, and cleavage. Crystal form, cleavage, and hardness are determined primarily by the crystal structure at the atomic level. Color and density are determined primarily by the chemical composition.
Is odor a chemical property?
Characteristics such as melting point, boiling point, density, solubility, color, odor, etc. are physical properties. Properties that describe how a substance changes identity to produce a new substance are chemical properties.
What are the chemical properties of bleach?
Bleaching. Household bleach is, in general, a solution containing 3–8% sodium hypochlorite, by weight, and 0.01–0.05% sodium hydroxide; the sodium hydroxide is used to slow the decomposition of sodium hypochlorite into sodium chloride and sodium chlorate.
What are the chemical properties of fluorine?
Fluorine is the most reactive and the most electronegative of all the elements. Fluorine is a pale yellow, diatomic, highly corrosive, flammable gas, with a pungent odor. It is the lightest halogen. It reacts violently with water to produce oxygen and the extremely corrosive hydrofluoric acid.
What are properties of bromine?
atomic number35melting point−7.2 °C (19 °F)boiling point59 °C (138 °F)specific gravity3.12 at 20 °C (68 °F)oxidation states−1, +1, +3, +5, +7
What are properties of liquids?
Liquids have the following characteristics: no definite shape (takes the shape of its container) has definite volume. particles are free to move over each other, but are still attracted to each other.
What is the six properties of matter?
The six physical properties are color, density, volume, mass, boiling point, and melting point.
What are the properties of matter Grade 8?
- Physical properties of matter are properties that can be measured or observed without matter changing to an entirely different substance. …
- Examples of physical properties of matter include melting point, color, hardness, state of matter, odor, and boiling point.
What are 5 examples of a physical change?
- Crushing a can.
- Melting an ice cube.
- Boiling water.
- Mixing sand and water.
- Breaking a glass.
- Dissolving sugar and water.
- Shredding paper.
- Chopping wood.
What are 3 examples of a chemical reaction?
Some examples of chemical reactions are combustion (burning), precipitation, decomposition and electrolysis.
What are physical and chemical properties?
Physical properties are those that can be observed without changing the identity of the substance. … Properties that describe how a substance changes into a completely different substance are called chemical properties. Flammability and corrosion/oxidation resistance are examples of chemical properties.
What is chemical change explain with example Class 6?
Chemcial Change When a new substance is formed after the change, it is called chemical change. Chemical changes cannot be reversed by simple physical means. Many chemical changes are irreversible. Examples: Burning of candle, rusting of iron, etc.
What are 4 physical changes?
Changes of state, for example, from solid to liquid or from liquid to gas, are also physical changes. Some of the processes that cause physical changes include cutting, bending, dissolving, freezing, boiling, and melting.
What are 10 physical changes?
- Vaporization.
- Smoke Formation. …
- Liquefaction Changes. …
- Freeze-drying. …
- Melting. …
- Freezing. …
- Dissolving. …
- Frost Formation. …
What are the 7 signs of a chemical reaction?
- Gas Bubbles Appear. Gas bubbles appear after a chemical reaction has occurred and the mixture becomes saturated with gas. …
- Formation of a Precipitate. …
- Color Change. …
- Temperature Change. …
- Production of Light. …
- Volume Change. …
- Change in Smell or Taste.
What are 4 facts about chemical changes?
- When ice melts it undergoes a physical change from solid to liquid. …
- Mixtures and solutions are different from chemical reactions as the molecules of the substances stay the same.
- Most cars get their power from an engine that uses a combustion chemical reaction.
What is a chemical change Class 7?
A change in which a substance undergoes a change in its chemical properties is called a chemical change. New substances are formed. For rusting, the presence of both oxygen and water (or water vapour) is essential. … A change in which one or more new substances are formed is called a chemical change.