Sulfuric acid 6% LDPE and HDPE at 20C°-50C° show little or no damage after 30 days of constant exposure. Sulfuric acid 20% LDPE and HDPE at 20C°-50C° show little or no damage after 30 days of constant exposure.
What plastic is resistant to sulfuric acid?
1. PTFE (or Teflon™) – PTFE, also commonly known as Teflon™, is one of the most acid resistant materials available and is commonly used in chemical and pharmaceutical lab applications.
Is HDPE plastic chemical resistant?
HDPE is a thermoplastic with good chemical resistance and corrosion resistance.
Is polyethylene resistant to sulfuric acid?
Due to its non-polar nature, the polyethylene resins (PEs) have an exceptionally high resistance to chemicals and other media. It is not affected by aqueous solutions of salts, acids and alkalis. … PEs are not resistant to strong oxidizing agents such as nitric acid, fuming sulfuric acid or halogens.Is plastic safe for sulfuric acid?
Storage Challenges. Sulfuric acid presents a serious storage challenge because it’s a very heavy chemical, especially at high concentrations. At 93-98% concentration it is nearly twice the weight of water. It’s also an aggressive chemical that oxidizes plastic and corrodes metals.
Is HDPE gasoline resistant?
Gasoline is a non-polar halo-carbon and contain a high amount of hydrogen which will permeate most polyofins (HDPE).
Is HDPE corrosion resistant?
High Density Polyethylene (HDPE) – HDPE is a corrosion resistant plastic that offers an increased working temperature range, and higher tensile strength when compared to Low-Density Polyethylene (LDPE).
What's the difference between HDPE and LDPE?
LDPE is softer and more flexible than HDPE. … HDPE is rigid and durable and offers greater chemical resistance. Its higher melting point (135° C) allows it to withstand higher temperatures than LDPE. Its more crystalline structure also results in greater strength and opacity of the material.Is HDPE chlorine resistant?
Not recommended for continuous use. Chlorine 10% in water HDPE and LDPE at 20C° shows little or no damage after 30 days. LDPE at 50C° shows damage and is not recommended.
Does acetone dissolve HDPE?While acetone will not dissolve HDPE, it can lead to material failure. If you leave acetone in a Nalgene bottle for long periods, it will eventually develop horizontal (i.e. circumferential) solvent-stress cracks at the level of the liquid-vapor interface, leading to failure when squeezed.
Article first time published onIs HDPE compatible with ammonia?
LDPE / HDPE at 20°C-50°C: little or no damage after 30 days. … LDPE / HDPE at 20°C-50°C: little or no damage after 30 days. Ammonia. LDPE / HDPE at 20°C-50°C: little or no damage after 30 days.
Is PVC resistant to sulfuric acid?
PVC is resistant to many alcohols, fats, oils and aromatic free petrol. It is also resistant to most common corroding agents including inorganic acids, alkalis and salts.
Is nylon resistant to sulfuric acid?
Nylon resins are stable in most inorganic chemicals including liquid ammonia, sulfurous acid, and others, but may be eroded with oxidants such as hydrogen peroxide and chlorine-based decolorants. Nylon resins are eroded in concentrated inorganic acids at room temperature, but fairly resistant to dilute acids.
What is resistant to sulfuric acid?
Titanium is chiefly known for its excellent corrosion resistance to a wide variety of oxidizing chemicals, such as nitric acid. It resists sulfuric in the presence of oxidizing salts, such as cupric and ferric sulfates.
What materials are resistant to sulfuric acid?
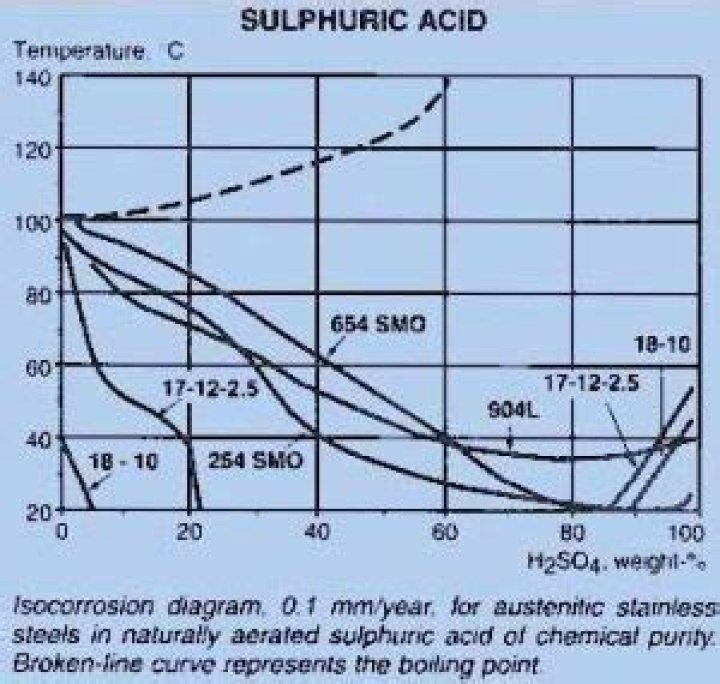
Chromium stainless steel, chromium nickel stainless steel and chromium nickel molybdenum stainless steel are similar, which have good corrosion resistance in room temperature concentrated sulfuric acid, and the corrosion rate also increases with the increase of temperature.
What kind of container can hold sulfuric acid?
The most common containers used for storing sulfuric acid solution quantities less than 10,000 gallons are high-density polyethylene (HDPE) tanks.
What dissolves HDPE?
HDPE and LDPE are easily soluble in decalin or decalin-cyclohexane (2:1 v/v) mixture at elevated temperatures (120-140 deg C). toluene, xylene, trichlorobenzene, tetralin and almost all high boiling point hydrocarbons. … Xylene is the best solvent for polyethylene. You need to heat it to fasten the dissolution.
Which polymer is most corrosion resistant?
Properties of PVC and CPVC are identical with the exception of temperature resistance where CPVC is clearly superior. Both PVC and CPVC are chemically and thermally bondable. PVC is the oldest corrosion resistant plastic. Both PVC and CPVC are available in sheet, rod, tubings as well as profiles.
Which acid is stored in plastic container?
Hydrofluoric acid must be stored in a tightly closed container made from either polyethylene, fluorocarbon, or lead.
Can I store petrol in HDPE?
The container should be of adequate strength in relation to its intended purpose, eg materials such as virgin high-density polyethylene (HDPE) would meet this need. Polyethylenes of uncontrolled composition and mixtures of different grades should not be used.
Can HDPE hold diesel?
Industrial-grade plastic drums are manufactured from HDPE, or high-density polyethylene, which is specifically designed to hold industrial goods. … Unfortunately, you can’t store diesel fuel in plastic drums forever. The diesel will react with the plastic polymers over time, even if the drum is produced from HDPE.
What type of plastic is used for gas tanks?
PLASTICS TECHNOLOGY. High-density polyethylene (HDPE) has been the resin of choice for plastic gas tanks, and production capacity has been on the increase.
Is HDPE compatible with ozone?
ABS plasticB – GoodHDPEB – GoodHypalon®A – ExcellentHytrel®C – FairKalrezA – Excellent
Is polycarbonate resistant to sulfuric acid?
Polycarbonate is resistant to mineral acids, many organic acids, oxidizing and reducing agents, neutral and acid salt solutions, many greases, waxes and oils, saturated, aliphatic and cycloaliphatic hydrocarbons and alcohols, with the exception of methyl alcohol.
Why is LDPE chemical resistant?
LDPE is defined by a density range of 0.910–0.940 g/cm3. It is not reactive at room temperatures, except by strong oxidizing agents, and some solvents cause swelling. … LDPE’s intermolecular forces are weaker, its tensile strength is lower, and its resilience is higher than HDPE.
Can you melt HDPE and LDPE together?
Unlike other polymers, HDPE and LDPE are miscible. They can be recycled together. However, to maintain their specific properties, it is better to separate them. LDPE has a branched structure.
Is HDPE stronger than LDPE?
LDPE’s long- and short-chain branches keep the material from packing tightly in its crystalline form. This gives it less tensile strength than HDPE, but greater ductility. … The molecules are tightly packed together during crystallisation, making HDPE dense and possessing higher resilience than LDPE.
Why is LDPE more flexible than HDPE?
The polymer chains of LDPE are highly branched compared to HDPE. This branching prevents the chains from stacking neatly beside each other, reducing the intermolecular forces of attraction. This results in a plastic that is softer and more flexible, but which also has lower tensile strength.
What chemical will dissolve polyethylene?
Polyethylene (other than cross-linked polyethylene) usually can be dissolved at elevated temperatures in aromatic hydrocarbons such as toluene or xylene, or in chlorinated solvents such as trichloroethane or trichlorobenzene.
What is the melting point of HDPE?
Thermoplastic materials become liquid at their melting point (110-130 degrees Celsius in the case of LDPE and HDPE respectively).
What is HDPE plastic made of?
High density polyethylene plastic is most commonly known and referred to as HDPE sheet plastic. This thermoplastic is made from a string of ethylene molecules (hence, the poly part of polyethylene), and is known for being both lightweight and strong.