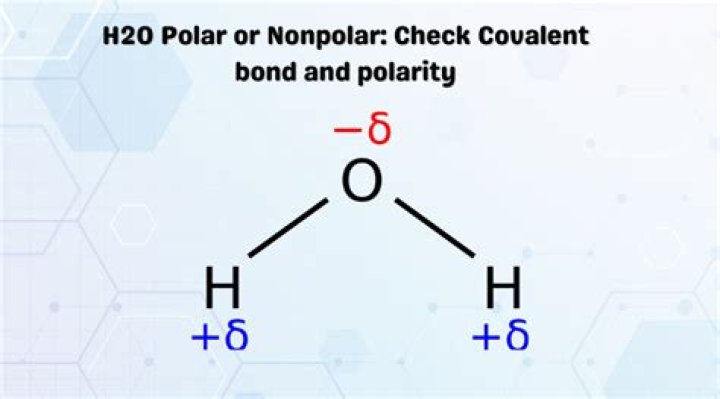
Water (H2O), like hydrogen fluoride (HF), is a polar covalent molecule. … The unequal sharing of electrons between the atoms and the unsymmetrical shape of the molecule means that a water molecule has two poles – a positive charge on the hydrogen pole (side) and a negative charge on the oxygen pole (side).
Is water a polar compound Why or why not?
Water (H2O) is a polar molecule because the electrons of the hydrogen atoms get “pulled” towards the electrons of the oxygen atom. This makes a region of positive charge on the hydrogen atoms and the negative charge on the other end of the molecule, which is the oxygen atom. This also allows for hydrogen bonding.
Why is h20 a polar bond?
Water (H2O) is polar because of the bent shape of the molecule. The shape means most of the negative charge from the oxygen on side of the molecule and the positive charge of the hydrogen atoms is on the other side of the molecule. This is an example of polar covalent chemical bonding.
How do you know that h2o is polar?
The unequal sharing of electrons gives the water molecule a slight negative charge near its oxygen atom and a slight positive charge near its hydrogen atoms. When a neutral molecule has a positive area at one end and a negative area at the other, it is a polar molecule.What do they mean by water is polar?
Water is a “polar” molecule, meaning that there is an uneven distribution of electron density. Water has a partial negative charge ( ) near the oxygen atom due the unshared pairs of electrons, and partial positive charges ( ) near the hydrogen atoms.
Why is water considered a polar molecule quizlet?
Water is considered a polar molecule because one side of the molecule is a different charge then the other side of the molecule. … The hydrogen atoms in water bond to the hydrogen atoms in the air, creating a greater surface tension than most other liquids.
How do you find the electronegativity difference in H2O?
Find the electronegativity difference between the two atoms. When two atoms are bonded together, the difference between their electronegativities can tell you about the qualities of their bond. Subtract the smaller electronegativity from the larger one to find the difference.
Why H2O is polar and CO2 is nonpolar?
Polar molecules have a non-zero net dipole moment. Both CO2 and H2O have two polar bonds. However the dipoles in the linear CO2 molecule cancel each other out, meaning that the CO2 molecule is non-polar. The polar bonds in the bent H2O molecule result in a net dipole moment, so H2O is polar.Is bei2 polar or nonpolar?
The molecular geometry of BeI2 is linear with symmetric charge distribution on the central atom. Therefore this molecule is nonpolar.
What forces does H2O have?Water has hydrogen bonds, dipole-induced dipole forces, and London dispersion forces.
Article first time published onIs H2O an atom?
For example, water (H2O) is a compound consisting of two hydrogen atoms bonded to an oxygen atom. The atoms within a compound can be held together by a variety of interactions, ranging from covalent bonds to electrostatic forces in ionic bonds.
Why is h20 a molecule?
Water is a molecule because it contains molecular bonds. Water is also a compound because it is made from more than one kind of element (oxygen and hydrogen). … Oxygen in the atmosphere is a molecule because it contains molecular bonds. It is not a compound because it is made from atoms of only one element – oxygen.
Is H2O an atom molecule element or compound?
The chemical formula for water is H2O, which means each molecule of water consists of one oxygen atom chemically bonded to two hydrogen atoms. Thus, water is a compound. It’s also a molecule, which is any chemical species formed by two or more atoms chemically bonded to each other.
What are polar particles?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. A diatomic molecule that consists of a polar covalent bond, such as HF, is a polar molecule.
Is water nonpolar molecule?
Water is a polar molecule. While the overall charge of the molecule is neutral, the orientation of the two positively charged hydrogens (+1 each) at one end and the negatively charged oxygen (-2) at the other end give it two poles.
Is NaCl polar?
Sodium Chloride (NaCl) which is an ionic compound acts as a polar molecule. Usually, the large difference in electronegativities in sodium and chlorine makes their bond polar.
What is the electronegativity P?
The first scale of electronegativity was developed by Linus Pauling and on his scale phosphorus has a value of 2.19 on a scale running from from about 0.7 (an estimate for francium) to 2.20 (for hydrogen) to 3.98 (fluorine).
How do you find the electronegativity?
On the periodic table, electronegativity generally increases as you move from left to right across a period and decreases as you move down a group. As a result, the most electronegative elements are found on the top right of the periodic table, while the least electronegative elements are found on the bottom left.
How do you determine polarity?
The terms “polar” and “nonpolar” usually refer to covalent bonds. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; if the result is between 0.4 and 1.7, then, generally, the bond is polar covalent.
What is H2O quizlet?
The scientific term for water is H2O. This means that in each water molecule there is two hydrogen atoms and one oxygen atom. … Water has a different charge in each of its ends. One is a positive charge and the other is a negative charge. These different charges cancel each other out and makes water a polar molecule.
Which properties of water are due to water being polar?
2 Answers. The polarity of water molecules means that molecules of water will stick to each other. This is called hydrogen bonding. Polarity makes water a good solvent, gives it the ability to stick to itself (cohesion), stick to other substances (adhesion), and have surface tension (due to hydrogen bonding).
Why is water also written as H2O quizlet?
The two elements that make up a molecule of water are hydrogen and oxygen; there are two hydrogen atoms and one oxygen atom, resulting in the chemical formula H2O. Hydrogen and oxygen bond together due to the number of electrons in their orbitals.
Is h20 tetrahedral?
Water has 4 regions of electron density around the central oxygen atom (2 bonds and 2 lone pairs). These are arranged in a tetrahedral shape. The resulting molecular shape is bent with an H-O-H angle of 104.5°.
Is BeI2 ionic or covalent?
Beryllium Fluoride (BeF2) is an ionic compound, whereas other halides like BeCl2, BeBr2 and BeI2 are highly covalent .
Is nh2cl polar or nonpolar?
The molecular geometry of NH2Cl is trigonal pyramidal with asymmetric charge distribution on the central atom. Therefore this molecule is polar. Chloramine on Wikipedia.
Why are CO2 bonds polar?
The carbon-oxygen double bonds in the linear CO2 molecule are polar (electronegativities: C = 2.5, O = 3.5). The electrons in each of the double bonds are drawn toward the oxygens, so both oxygen atoms have a partial negative charge.
What is the polarity of CO2?
Carbon dioxide, which has the chemical formula CO2, is non-polar. It contains two polar bonds that are arranged symmetrically. Carbon forms a double bond with each oxygen atom.
Why CO2 is non polar But SO2 is polar?
CO2 is linear so its dipoles cancel. However, since SO2 is bent, the dipoles do not cancel and the molecule is polar.
What intermolecular forces are present in H2O and H2O?
The H2O water molecule is polar with intermolecular dipole-dipole hydrogen bonds. As the water molecules attract each other and form bonds, water displays properties such as high surface tension and a high heat of vaporization.
What predominant intermolecular force is in H2O?
Option b (hydrogen bonding) is the correct answer. Hydrogen bonding is generally the strongest intermolecular force and is found in compounds that have N-H, O-H or F-H bonds in their structure. As water is composed of two O-H bonds, it is capable of hydrogen bonding.
What intermolecular forces are present in HCN?
(d) HCN is a linear molecule; it does have a permanent dipole moment; it does contain N, however the nitrogen is not directly bonded to a hydrogen. Therefore dispersion forces and dipole-dipole forces act between pairs of HCN molecules.