Electronegativity DifferenceType of Bond0.0 to 0.5Nonpolar Covalent0.6 to 1.7Polar Covalent> 1.7Ionic
Is 1.7 A ionic?
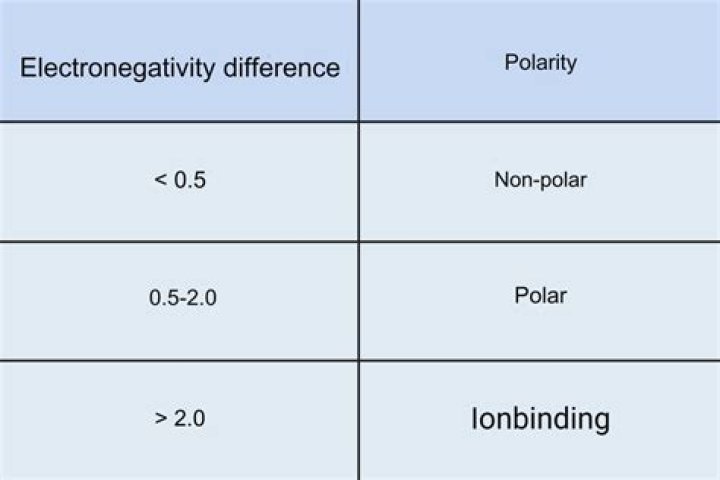
If the electronegativity difference is more than 1.7, the bond will have an ionic character. If the electronegativity difference is between 0.4 and 1.7, the bond will have a polar covalent character. Lastly, if the electronegativity difference is less than 0.4, the bond will have a nonpolar covalent character.
Is 1.6 polar or ionic?
If the ΔEN is between 0.5 and 1.6 (some use 1.7), the bond is considered polar covalent. If the ΔEN is greater than 2.0, then the bond is ionic. If the ΔEN is between 1.6 and 2.0, and a metal is involved, then the bond is considered ionic. If only nonmetals are involved, the bond is considered polar covalent.
Is 1.7 polar covalent?
Polar Covalent Bonds A bond in which the electronegativity difference between the atoms is between 0.4 and 1.7 is called a polar covalent bond.Is 1.5 polar or ionic?
Electronegativity DifferenceType of Bond Formed0.0 to 0.2nonpolar covalent0.3 to 1.4polar covalent> 1.5ionic
Is 1.9 polar or ionic?
Electronegativity DifferenceBond Type0nonpolar covalent0–0.4slightly polar covalent0.4–1.9definitely polar covalent>1.9likely ionic
Is 1.8 an ionic?
Identical atoms have zero EN difference, hence nonpolar. Answer d: The EN difference is greater than 1.8, hence ionic.
Is OO polar or nonpolar?
An O-O bond is covalent, since the electrons are shared between the two oxygen atoms. Moreover, the bond is nonpolar, since the atoms linked together…When the ΔEN 1.7 The type of bond is?
ΔENBondingBond Example1.0 – 1.3Moderately polar covalent bondC-O, S-O1.4 – 1.7Highly polar covalent bondH-O1.8 – 2.2Slightly ionic bondH-F2.3 – 3.3Highly ionic bondNa+ F-
Is O O polar or nonpolar?In ozone (O3) molecules, the two O−O bonds are nonpolar (there is no electronegativity difference between atoms of the same element).
Article first time published onIs 1.6 polar or nonpolar?
For a bond to be polar, the electronegativity difference between the two elements needs to be between 0.5 to 1.6. If the electronegativity difference is less than 0.5, the bond is nonpolar. Any more than 1.6 and the molecules become charged ions and form ionic bonds instead.
What electronegativity difference is ionic?
Ionic bonds form between atoms with an electronegativity difference > 1.7.
What is the polarity of CL?
λF (nm)ΦF (in AN)DTT3331.0 × 10−3DTP3377.7 × 10−5
What is the significance of an electronegativity difference of 1.7 between 2 atoms apex?
the difference between the electronegativity of the two atoms in a bond will determine whether the bond is ionic or covalent. If the difference in electronegativity is greater than 1.7, the bond is considered ionic.
How do you know a molecule is polar?
The terms “polar” and “nonpolar” usually refer to covalent bonds. To determine the polarity of a covalent bond using numerical means, find the difference between the electronegativity of the atoms; if the result is between 0.4 and 1.7, then, generally, the bond is polar covalent.
Is 2.0 polar or ionic?
If the electronegativity difference between the two atoms is between 0.5 and 2.0, the atoms form a polar covalent bond. If the electronegativity difference between the atoms is greater than 2.0, the bond is ionic.
What considered polar?
Although there are no hard and fast rules, the general rule is if the difference in electronegativities is less than about 0.4, the bond is considered nonpolar; if the difference is greater than 0.4, the bond is considered polar.
Is sih polar?
The difference in electronegativity for both bonds is approximately 0.3, but the C-H bond is considered to be nonpolar covalent, while the Si-H bond is considered to be polar covalent. Because silicon is a larger atom than carbon, it will also have a larger electron cloud.
Is 1.9 polar or non polar?
Obviously there is a wide range in bond polarity, with the difference in a C-Cl bond being 0.5 — considered just barely polar — to the difference the H-O bonds in water being 1.4 and in H-F the difference is 1.9.
Is ch4an ionic compound?
Each ball in the gas diagram above represents one methane molecule. Is CH4 ionic or covalent? … Methane, CH4, is a covalent compound with exactly 5 atoms that are linked by covalent bonds.
Which compound is more polar?
The shared electrons of the covalent bond are held more tightly at the more electronegative element creating a partial negative charge, while the less electronegative element has a partial positive charge, . The larger the difference in electronegativity between the two atoms, the more polar the bond.
What is ΔEN?
The absolute value of the difference in electronegativity (ΔEN) of two bonded atoms provides a rough measure of the polarity to be expected in the bond and, thus, the bond type. When the difference is very small or zero, the bond is covalent and nonpolar. When it is large, the bond is polar covalent or ionic.
What accounts for the polarity of compounds?
The polarity of a compound depends on how the atoms within the compound are attracted to each other. This attraction can create a difference in electronegativity if one atom or molecule has more “pull” than another and make the molecule polar.
Is OO a covalent bond?
When the two bonded atoms are atoms of the same element, the electronegativity difference is 0, and the bond is nonpolar covalent. So the bond character of the O-O bond in hydrogen peroxide, H2O2 , is nonpolar covalent, because the electronegativity difference is 0.
What kind of bond is Cl O?
Chlorine and Oxygen both are non metals having 7 and 6 electrons in their outer most orbit respectively. So the type of bond that they follow is Covalent Bond which is based on sharing of electrons.
Is o3 polar?
The shape of ozone molecule is bent and the central oxygen has a positive charge, while the single bonded oxygen has a negative charge. Because of the molecule’s shape, the dipole moments do not cancel, so the ozone molecule is polar.
What are examples of polar molecules?
- Water (H2O) is a polar molecule. …
- Ethanol is polar because the oxygen atoms attract electrons because of their higher electronegativity than other atoms in the molecule. …
- Ammonia (NH3) is polar.
- Sulfur dioxide (SO2) is polar.
- Hydrogen sulfide (H2S) is polar.
What are H bonds?
hydrogen bonding, interaction involving a hydrogen atom located between a pair of other atoms having a high affinity for electrons; such a bond is weaker than an ionic bond or covalent bond but stronger than van der Waals forces.
Is CH3CN ionic or covalent?
These measures show that the CuCN and CuNC bonding is dominantly ionic while the bonding for CH3CN and CH3NC is dominantly covalent. For the CH3 containing molecules, there is participation of the cyanide 2 pi orbital in the bond however, it is not involved in the ionic bond with Cu.
Is CCl4 polar or nonpolar?
CCl4 that is carbon tetrachloride is nonpolar because all the four bonds are symmetrical, and they are they extended in all the directions. This makes it easy for the dipole moments in each direction to cancel out.
What's a polar molecule?
A polar molecule is a molecule in which one end of the molecule is slightly positive, while the other end is slightly negative. A diatomic molecule that consists of a polar covalent bond, such as HF, is a polar molecule. … A molecule with two poles is called a dipole (see figure below). Hydrogen fluoride is a dipole.