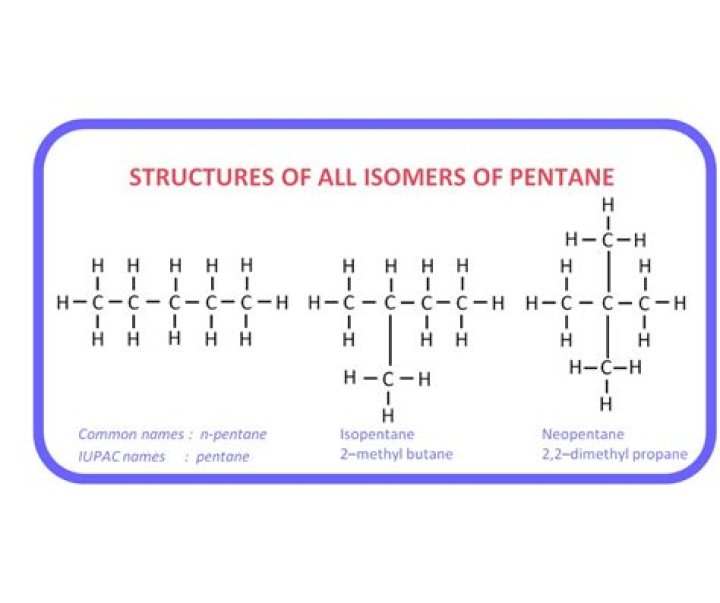
There are three structural isomers of butene.
How many chain isomers does butene have?
butene, also called Butylene, any of four isomeric compounds belonging to the series of olefinic hydrocarbons. The chemical formula is C4H8. The isomeric forms are 1-butene, cis-2-butene, trans-2-butene, and isobutylene.
How many isomers does but-1-ENE have?
Notice that butene has two different forms called isomers .
What is the chain isomer of but-1-Ene?
namestructural formulamolecular formulabut-1-ene (1-butene)H | H | H | C = C – C – C -H | H | H | H | HC4H8How many isomers are possible for but-2-ene?
2-Butene is an acyclic alkene with four carbon atoms. It is the simplest alkene exhibiting cis/trans-isomerism (also known as (E/Z)-isomerism); that is, it exists as two geometric isomers cis-2-butene ((Z)-2-butene) and trans-2-butene ((E)-2-butene).
Are but-1-ene and but-2-ene position isomers?
Isomers with a difference in the position of multiple bond/functional group/alkyl substituent on the same carbon skeleton (parent chain) is known as position isomers. Butene exhibits positon isomerism as but-1-ene and but-2-ene differ in the position of double bond.
Are butane and but-1-ene isomers?
NamesRefractive index (nD)1.3962Viscosity7.76 PaHazardsGHS pictograms
What kind of isomers are but-1-Ene and 2 ene?
But-1-ene and but-2-ene are examples of position isomerism.Is Prop 1 ENE correct?
4 of the current version of Nomenclature of Organic Chemistry – IUPAC Recommendations and Preferred Names 2013 (Blue Book). Ignoring the rules for omission of locants, the complete systematic name of the parent compound would be ‘prop-1-ene’. According to Rule P-14.3. 4.2, however, the locant ‘1‘ is omitted.
What does the 1 mean in but-1-Ene?Alkenes are hydrocarbons which contain carbon-carbon double bonds. Thus, the word “1-butene” indicates a chain of four carbons, with a double bond between carbons 1 and 2; the word “2-butene” indicates a chain of four carbons, with a double bond between carbons 2 and 3. …
Article first time published onIs but-1-ene saturated or unsaturated?
Number of CarbonSubstanceType3propyneunsaturated4butanesaturatedbut-1-eneunsaturatedbut-1-yneunsaturated
Is but 2 ene saturated?
2-butene appears as a colorless liquefied petroleum gas. Asphyxiate gas. Flammability limits in air 1.8-9.7% by volume.
How does but-2-ene show geometrical isomerism?
Since isomers have the same molecular formula the difference in their properties might be due to different modes of combination of arrangement of atoms within the molecule. … Since the above compound (but-2-ene) has double bonds and is an alkene therefore is a geometrical isomer.
Is but-2-ene a geometric isomer?
The most likely example of geometric isomerism you will meet at an introductory level is but-2-ene. In one case, the CH3 groups are on opposite sides of the double bond, and in the other case they are on the same side. Geometric isomers can only occur where there is restricted rotation about a bond.
Do geometric isomers exist for but-1-Ene?
but-1-ene. ⚠ Does not have geometrical isomers, even though it has a C=C bond! One of the C atoms in this bond has two identical groups (H atoms) attached.
What type of polymerisation does but-1-ene undergo?
In a rather low percentage, but-1-ene is used as one of the co-monomers, along with other α-olefins, in the production of high-density polyethylene (HDPE) and linear low-density polyethylene (LLDPE). Butyl rubber is made by cationic polymerisation of isobutylene with between 2 and 7% isoprene.
How can you convert but-1-ene to 1 Iodobutane?
i.e. the attack of HBr on but-1-ene in the presence of peroxide gives 1-bromobutane as the intermediate which on treatment with NaI+Acetone gives 1-iodobutane as the final product and NaBr as the by-product.
What is the major product of the reaction of HBr with but-1-ene?
For instance, the reaction between HBr and 1-butene that has just been distilled usually yields the Markovnikov product 2-bromobutane. Astonishingly, if 1-butene is exposed to air prior to the experiment, the addition of HBr predominantly results in the anti-Markovnikov product 1-bromobutane.
Is there a but 3 Ene?
But-3-ene hydrochloride | C4H9Cl – PubChem.
Is butene and but 1 ene the same?
There are no differences. Synonyms for 1-butene (C2H5CH=CH2) are Synonyms: but-1-ene, n-butene, butene-1, ethyl ethylene, alpha-butylene, alpha-butene, n-butylene.
What is the functional group in but 2 ene?
Chemical classGroupExampleAlkeneAlkenyl(E)-But-2-eneAlkyneAlkynylBut-2-yneAlleneAllenylPenta-2,3-dieneCumuleneCumulenylHexa-2,3,4-triene
How will you bring about the following conversion but 1 ene to but-2-ene?
Conversion of but-1-ene to but-2-ene is by catalytic isomerisation. But-1-ene undergoes isomerisation to but-2-ene on a nickel catalyst in the presence of hydrogen atoms. … We then add alcoholic KOH to get but-2-ene.
What is positional isomer?
Position isomers (also positional isomers or regioisomers) are structural isomers that can be viewed as differing only on the position of a functional group, substituent, or some other feature on a “parent” structure.
What is c5h10 classified as?
Cyclopentane is a cycloalkane that consists of five carbons each bonded with two hydrogens above and below the plane. The parent of the class of cyclopentanes. It has a role as a non-polar solvent. It is a member of cyclopentanes, a cycloalkane and a volatile organic compound. ChEBI.
Where is propylene found?
Propylene is obtained during the refining of gasoline. But it can also be produced by splitting, cracking and reforming hydrocarbon mixtures. Propylene is an attractive alternative to propane for heating and cutting due to its superior combustion performance.
Is E but-2-ene symmetrical?
The product produced contains a chlorine atom. And the carbon-carbon double bond of the alkene is now a carbon-carbon single bond. … But if we flip the bottom product over, we can see that the two product molecules are in fact the same. This has occurred because but-2-ene is a symmetrical molecule.
How is propylene made?
The propylene molecule is produced as a co-product of ethylene production through the steam cracking (steam pyrolysis) of hydrocarbon feedstocks. Feedstocks used for steam cracking range from ethane to naphtha and gas oils. Propylene is also produced as a by-product of petroleum refining.
Are but 1 Ene and 2 Methylprop 1 ENE position isomers?
Complete answer: $But – 1 – ene$ and $2 – methylprop – 1 – ene$ are not position isomers because as we know position isomers have the same molecular formula but differ in position of the functional group of $C = C$ .
What type of isomers are propan 1 ol and propan 2?
Functional isomers are defined as the isomers that have the same molecular formula but they are attached to different functional groups. In this question, both the chemical compounds, that are, Propan$-1-$ol and Propan$-2-$ol contain the same hydroxy group.
Which of the following can show positional isomerism?
chair and boat forms of cyclohexane.
Is it ethene or ETH 1 Ene?
Number of carbon atoms:15Prefix:methpent