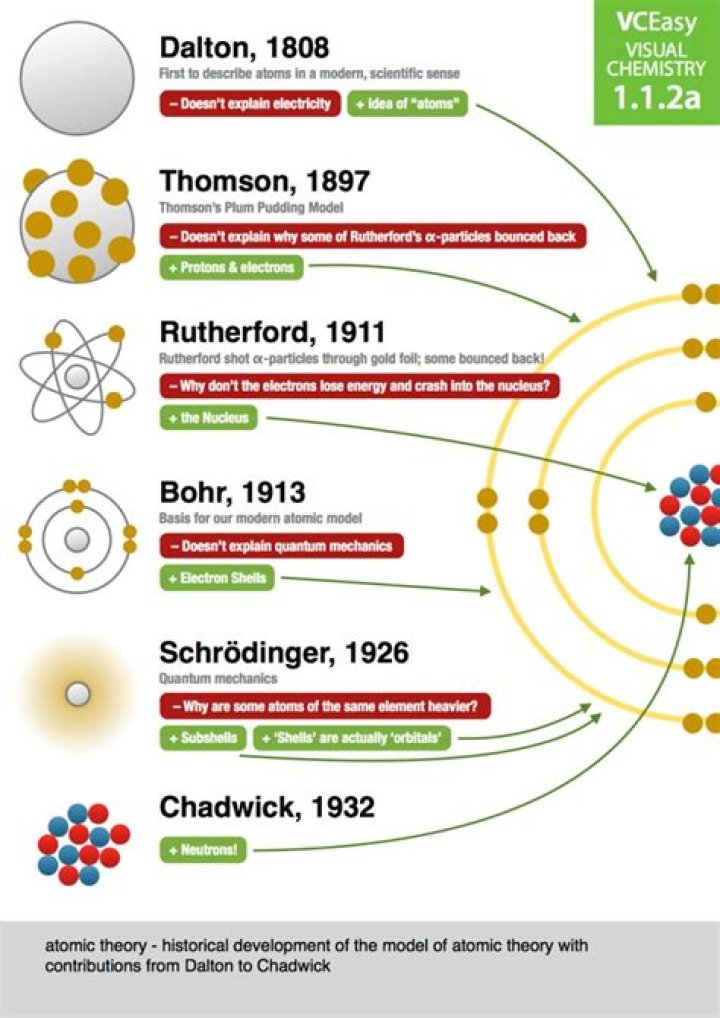
The Dalton model has changed over time because of the discovery of subatomic particles .
How did Dalton's model changed over time?
The Dalton model has changed over time because of the discovery of subatomic particles .
How has the model of the atom has changed over time?
This atomic model has changed over time. Scientists used the model to make predictions. Sometimes the results of their experiments were a surprise and they did not fit with the existing model. Scientists changed the model so that it could explain the new evidence.
How was Dalton's model modified?
On the basis of new experimental facts, Dalton’s atomic theory has been modified as: … Atoms of same element may not have same atomic mass. For example, some atoms of chlorine have atomic mass 35 amu while others have atomic mass 37 amu. Such atoms of same element having different atomic masses are called isotopes.What model replaced Dalton's model?
Thomson proposed that the electrons are embedded into a positively charged sphere so that the atom as a whole is electrically neutral. That could explain charge neutrality, and chemical bond formation to some extent.
What were the 5 most important points in Dalton's atomic theory?
Terms in this set (5) Compounds are composed of atoms of more than 1 element. The relative number of atoms of each element in a given compound is always the same. Chemical reactions only involve the rearrangement of atoms. Atoms are not created or destroyed during chemical reactions.
What did Dalton discover about the atom BBC Bitesize?
Dalton’s model (1803) the atoms of a given element are identical to each other. the atoms of different elements are different from one another.
How did Dalton discover the atom?
In 1803 Dalton discovered that oxygen combined with either one or two volumes of nitric oxide in closed vessels over water and this pioneering observation of integral multiple proportions provided important experimental evidence for his incipient atomic ideas.What did Dalton contribute to the atom?
Dalton’s atomic theory proposed that all matter was composed of atoms, indivisible and indestructible building blocks. … Dalton also postulated that chemical reactions resulted in the rearrangement of the reacting atoms.
How did John Dalton find this new information?Dalton’s experiments on gases led to his discovery that the total pressure of a mixture of gases amounted to the sum of the partial pressures that each individual gas exerted while occupying the same space. In 1803 this scientific principle officially came to be known as Dalton’s Law of Partial Pressures.
Article first time published onWhy do scientific models change over time?
Scientists used the model to make predictions about their experiments. Often the data did not agree with their predictions. This meant that the model had to be changed. The modern atomic model is the result of many scientists building on each other’s work.
How are the Dalton and Thomson models different?
Note: The basic difference between the two models lies in the fact that Dalton proposed that an atom was indivisible and indestructible whereas Thomson worked on the existence of subatomic particles inside an atom and their arrangements i.e., he considered an atom to be a divisible quantity unlike Dalton.
How are Dalton's model and electron cloud models of the atom similar How are they different?
Chadwick found that the nucleus contained positive protons and neutral neutrons. How are Dalton’s model (see figure on previous page) and electron cloud models of the atom similar? How are they different? They both are a blur containing all of the electrons somewhere within it.
What happened in 1932 to change the idea of atoms?
By 1920, physicists knew that most of the mass of the atom was located in a nucleus at its center, and that this central core contained protons. In May 1932 James Chadwick announced that the core also contained a new uncharged particle, which he called the neutron.
How did the model of the atom change as new evidence was discovered?
New experimental evidence may lead to a scientific model being changed or replaced. Before the discovery of the electron, atoms were thought to be tiny spheres that could not be divided. The discovery of the electron led to the ‘plum-pudding model’ of the atom.
How did this contradict Dalton's model of the atom?
The modern atomic theory contradicts dalton’s atomic theory : Atoms are divisible into protons, neutrons, electrons. … Atoms of different elements have the same properties, such atoms are isobars. Atoms can combine in any ratio and not just whole numbers.
How did Bohr adapt the nuclear model?
In 1913, Niels Bohr revised Rutherford’s model by suggesting that the electrons orbited the nucleus in different energy levels or at specific distances from the nucleus. … Therefore, electrons cannot be arranged at random, but they must have fixed levels of energy within each type of atom.
What are the 4 major ideas of Dalton's atomic theory?
1) All matter is made of atoms. Atoms are indivisible and indestructible. 3) Compounds are formed by a combination of two or more different kinds of atoms. 4) A chemical reaction is a rearrangement of atoms.
Which Dalton theory is incorrect?
Drawbacks of Dalton’s Atomic Theory The indivisibility of an atom was proved wrong: an atom can be further subdivided into protons, neutrons and electrons. However an atom is the smallest particle that takes part in chemical reactions. According to Dalton, the atoms of same element are similar in all respects.
What are the main ideas in Dalton's atomic theory quizlet?
What are the main ideas in Dalton’s atomic theory? Atoms combine in simple, whole number ratios to form compounds. All atoms of a given element have the same mass and other properties that distinguish them from the atoms of other elements. Atoms of one element cannot change into atoms of another element.
What is the atomic theory proposed by John Dalton What changes have taken place in the theory during the last two centuries?
what changes have taken place in the theory during the last two centuries? All matter is made of atom and atoms are indestructible. An atom of an element is identical in mass and property. Compounds are formed by the combination of two or more atoms.
Did Dalton believe that atoms could be created or destroyed?
Dalton proposed his atomic theory in 1804. … Atoms cannot be subdivided, created, or destroyed. Atoms of different elements can combine in simple whole number ratios to form chemical compounds.
What contribution did John Dalton make to atomic theory quizlet?
John Dalton’s Atomic Theory included these statements. (1) All matter is composed of tiny, indivisible particles, called atoms, that cannot be destroyed or created. (2) Each element has atoms that are identical to each other in all of their properties.
What was Bohr experiment?
The Bohr model shows the atom as a small, positively charged nucleus surrounded by orbiting electrons. Bohr was the first to discover that electrons travel in separate orbits around the nucleus and that the number of electrons in the outer orbit determines the properties of an element.
What did Bohr contribute to the atom?
What was Niels Bohr’s most important discovery? Niels Bohr proposed a model of the atom in which the electron was able to occupy only certain orbits around the nucleus. This atomic model was the first to use quantum theory, in that the electrons were limited to specific orbits around the nucleus.
Why was the Dalton model proposed?
Dalton hypothesized that the law of conservation of mass and the law of definite proportions could be explained using the idea of atoms. He proposed that all matter is made of tiny indivisible particles called atoms, which he imagined as “solid, massy, hard, impenetrable, movable particle(s)”.
How did John Dalton's work contribute to the discovery of new elements?
John Dalton developed a crude method for measuring the masses of the elements in a compound. His law of multiple proportions states that when two elements form more than one compound, masses of one element that combine with a fixed mass of the other are in a ratio of small whole numbers.
What discoveries did John Dalton make?
Although a schoolteacher, a meteorologist, and an expert on color blindness, John Dalton is best known for his pioneering theory of atomism. He also developed methods to calculate atomic weights and structures and formulated the law of partial pressures.
Can scientific models be updated edited or changed?
Scientific models are not a replacement for experimentation. They can be used in conjunction with experimentation to further understanding of a concept, event, or process. They are based on current scientific knowledge and may have to be changed when new discoveries are made.
What would cause models to change?
A model needs to be changed when observations are not matching what the model predicts in a given situation. That does not mean, necessarily, that the initial model is bad, it can simply mean the assumptions under which the model is working are not respected.
Which best describes why scientific models change over time 2 points?
Which best describes why scientific models change over time? As technology advances, new experiments often expose problems in accepted theories.