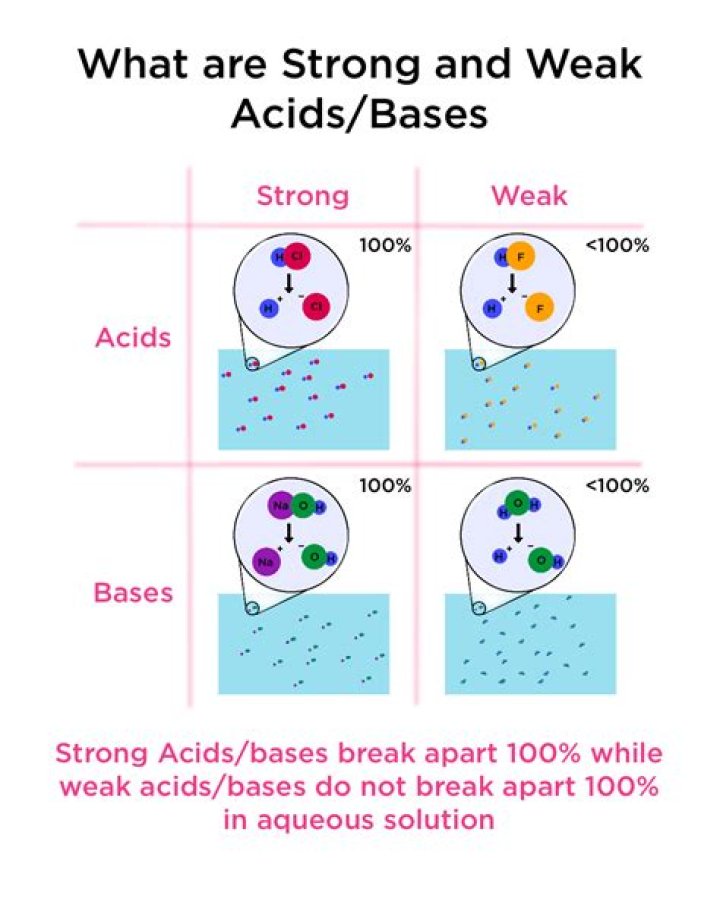
In any acid-base reaction, the equilibrium will favor the reaction that moves the proton to the stronger base. This equilibrium constant is referred to as the ion-product constant for water, Kw. … In pure water, some molecules act as bases and some as acids. This is referred to as autoionization.
How does acid affect equilibrium?
If hydrochloric acid was added to the equilibrium mixture, both hydrogen ions (H +) and chloride ions (Cl -) are being added. Hydrogen ions are on the right hand side of the equilibrium, therefore the equilibrium will shift to the left hand side to compensate, resulting in a higher concentration of reactants.
How does adding an acid-base affect an indicators equilibrium system?
In this reaction, adding acid shifts the indicator equilibrium to the left. Conversely, adding a base shifts the indicator equilibrium to the right. In the case of the indicator methyl orange, the HIn is colored red and the ionized In– form is yellow.
How does adding a base affect equilibrium?
Adding more OH- ions increases the pH, making the substance more basic. For reactions that have OH- ions in the product side: Increasing the pH will increase the number of OH- ions, so the equilibrium will shift to the left.Are acid base reactions equilibrium?
Acid base reactions are reversible and therefore equilibrium reactions. With acid base reactions we focus on the extent to which theACID (reactant) is deprotonated to the conjugate acid (product). Therefore, as the equivalent to the equilibrium constant, we look at the Ka.
What factors affect equilibrium?
Changes in concentration, temperature, and pressure can affect the position of equilibrium of a reversible reaction. Chemical reactions are equilibrium reactions. Equilibrium occurs when a certain proportion of a mixture exists as reactants and the rest exits as products.
What happens when acid reacts with base write the name of reaction give the example?
Explanation: The reaction between an acid and a base is known as a neutralisation reaction. Often when an acid and base react a salt and water will be formed. … Hydrochloric acid reacts with sodium hydroxide to form sodium chloride (a salt) and water.
What factors affect the equilibrium constant?
The only thing that changes an equilibrium constant is a change of temperature. The position of equilibrium is changed if you change the concentration of something present in the mixture.What affects equilibrium position?
Concentration, pressure, and temperature all affect the equilibrium position of a reaction, and a catalyst affects reaction rates. However, only temperature affects the value of Kc.
How do you determine acid base equilibrium?- Identify the acid and the conjugate acid of the base: …
- Look up the pKa values of the acid the conjugate acid: …
- Write the reaction equation pointing the equilibrium position with a longer arrow:
How do acids and bases affect the environment?
Acid rain increases acidity in bodies of water like streams and lakes. This alone damages aquatic organisms; however, an increase in acidity also causes aluminum that is found in soil to be leached into bodies of water. The combined impact of aluminum and acidity causes damage to aquatic ecosystems.
What is potentiometric acid base titration?
Potentiometric titration is a technique similar to direct titration of a redox reaction. It is a useful means of characterizing an acid. No indicator is used; instead the potential is measured across the analyte, typically an electrolyte solution.
Do strong acids form equilibrium?
This phenomenon is called the leveling effect: any species that is a stronger acid than the conjugate acid of water (H3O+) is leveled to the strength of H3O+ in aqueous solution because H3O+ is the strongest acid that can exist in equilibrium with water.
What is the equilibrium constant for the acid-base reaction between ammonia and acetic acid?
The equilibrium constant for the process is. Keq=10−pKa of acetic acid (reactant) 10−pKa of ammonium ion (product) =10−4.710−9.3=104.6. … We see that although acetic acid is a weak acid and ammonia is a weak base, the acid-base reaction between them is virtually complete.
Why do acid base reactions favor the weaker acid?
In an equilibrium-controlled acid–base reaction, the equilibrium position always favours the formation of the weaker acid and the weaker base. This is because the weaker acid and the weaker base are the most stable species due to their lower potential energies.
Why are acid base reactions important?
Homeostasis, the temperature and chemical balances in our bodies, is maintained by acid-base reactions. For example, fluctuations in the pH, or concentration of hydrogen ions, of our blood is moderated at a comfortable level through use of buffers.
What happens when acid reacts with a base explain by taking the example of HCl and NaOH What is the special name of such reaction?
When hydrochloric acid reacts with sodium hydroxide solution, then a neutralisation reaction takes place to form sodium chloride and water. Such a reaction is termed as neutralisation reaction.
What happens when acid react with acid?
The more the number of hydrogen ions liberated by an acid, the stronger is the acid. The strong acids have a very low pH of 1 – 2. When we mix two acids of the same strength, we can see that no reaction occurs. It is because the resultant will be neutral and there will be no change in the pH.
What causes shift in equilibrium?
If the concentration of a substance is increased, the reaction that consumes that substance is favored, and the equilibrium shifts away from that substance. If the concentration of a substance is decreased, the reaction that produces that substance is favored, and the equilibrium shifts toward that substance.
Which factor does not affect the equilibrium?
change in pressure or concentration of reactants does not affect equilibrium.
What is the most important factor that affects chemical equilibrium?
It states that changes in the temperature, pressure, volume, or concentration of a system will result in predictable and opposing changes in the system in order to achieve a new equilibrium state.
What factor is equilibrium constant independent?
Characteristics of Equilibrium Constant: It is independent of the initial concentrations of the reacting species. It changes with the change in the temperature. It depends on the nature of the reaction. It is independent of the change of pressure, volume and concentrations of the reactants and products.
How the concept of acids and bases predict the feasibility of a reaction?
Answer: Whether or not a reaction (or other physical change) is feasible depends on the sign of ΔG. If ΔG is positive, then the reaction isn’t feasible – it can’t happen. For a reaction to be feasible, ΔG has to be negative.
Does equilibrium favor the stronger base?
Weak acids and bases are lower in energy than strong acids and bases, and because equilibria favor the reaction side with the lowest-energy species, acid-base reactions will go to the side with the weakest acids and bases. As a rule, the equilibrium of a reaction will favor the side with weaker acids and bases.
What are the effects of acids in the environment?
Acid rain and fog also damage forests, especially those at higher elevations. The acid deposits rob the soil of essential nutrients such as calcium and cause aluminum to be released in the soil, which makes it hard for trees to take up water. Trees’ leaves and needles are also harmed by acids.
How does acid affect the environment?
Acid rain leaches aluminum from the soil. That aluminum may be harmful to plants as well as animals. Acid rain also removes minerals and nutrients from the soil that trees need to grow.
How do base affect the environment?
Bases can cause the soil to disrupt the growth of plants. Plants grow well when grown in soil and rocks that contain bases, such as Alkaline.
What is the advantage of potentiometric titration?
Advantages of potentiometric titration: It is an economical titration method. It requires a small quantity of substances. There is no need to use indicators for potentiometric titration. The titrations results are accurate as no colour indicators are involved.
What is end point in potentiometric titration?
Introduction. Potentiometric titration belongs to chemical methods of analysis in which the endpoint of the titration is monitored with an indicator electrode that records the change of the potential as a function of the amount (usually the volume) of the added titrant of exactly known concentration.
What do we mean by potentiometric titration and give one advantage of it?
Potentiometric titration is used for measuring the change in the electrical potential when a neutralizing agent (titrant) is added to a chemical solution. The main advantage of using Potentiometric titration method is that it is an inexpensive method. It uses small quantities of substances.