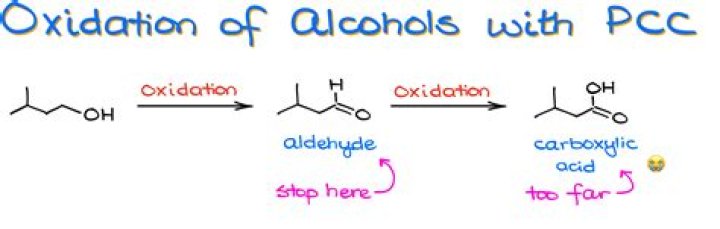
A common reagent that selectively oxidizes a primary alcohol to an aldehyde (and no further) is pyridinium chlorochromate, PCC. E.g. Tertiary Alcohols These are resistant to oxidation because they have no hydrogen atoms attached to the oxygen bearing carbon (carbinol carbon).
Can you oxidize a tertiary alcohol?
No further oxidation is seen except under very stringent conditions. Tertiary alcohols cannot be oxidized at all without breaking carbon-carbon bonds, whereas primary alcohols can be oxidized to aldehydes or further oxidized to carboxylic acids.
Which of the following alcohol is not oxidized by PCC?
Option A is nothing but phenol, which cannot be oxidized by using PCC. Option A is the correct one. Note: As PCC is a weak oxidizing agent, it cannot oxidize the primary alcohols directly to carboxylic acids but oxidizes primary alcohols to aldehydes only.
What can PCC oxidize?
Explanation: PCC can be used to oxidize primary alcohols into aldehydes, or secondary alcohols into ketones. The starting material shown is a secondary alcohol, so the product will be a ketone (a carbonyl ( ) group where the carbonyl carbon is also attached to two other carbons).Why is PCC used for alcohol oxidation?
Pyridinium chlorochromate (PCC) is a milder version of chromic acid. PCC oxidizes alcohols one rung up the oxidation ladder, from primary alcohols to aldehydes and from secondary alcohols to ketones. … This is not a concern with ketones because there is no H directly bonded to C.
Can KMNO4 oxidize a tertiary alcohol?
Yes, that’s right. Tertiary alcohols readily undergo elimination to yield alkenes, then the KMNO4 reacts with the alkene to give syn dihydroxylation.
Why can't you oxidize a tertiary alcohol?
Tertiary alcohols (R3COH) are resistant to oxidation because the carbon atom that carries the OH group does not have a hydrogen atom attached but is instead bonded to other carbon atoms. … Therefore tertiary alcohols are not easily oxidized.
Can PCC oxidize ketone?
Oxidation of alcohols PCC is used as an oxidant. In particular, it has proven to be highly effective in oxidizing primary and secondary alcohols to aldehydes and ketones, respectively.What happens when alcohol reacts with PCC?
Formation of Aldehydes using PCC PCC oxidizes alcohols one rung up the oxidation ladder, from primary alcohols to aldehydes and from secondary alcohols to ketones. Unlike chromic acid, PCC will not oxidize aldehydes to carboxylic acids.
What does h2cro4 do to an alcohol?Chromic acid (H2CrO4) oxidizes alcohols in aqueous solutions of sodium dichromate. It reacts with alcohols to form a chromic ester in which the alcohol oxygen atom bridges the carbon and chromium atoms. Thus, the ester forms by nucleophilic attack of the alcohol’s oxygen atom on the chromium atom.
Article first time published onWhich of the following is a tertiary alcohol?
Thus 2-methylbutan-2-ol is a tertiary alcohol.
Which of the following Cannot oxidize ethanol?
Tertiary alcohols cannot be oxidized.
What is a tertiary alcohol structure?
A tertiary alcohol is a compound in which a hydroxy group, ‒OH, is attached to a saturated carbon atom which has three other carbon atoms attached to it.
What does PCC and DCM do?
PCC is soluble in many organic solvents, and especially dichloromethane at room temperature has been used in most cases, whereas DMF promotes the over-oxidation of primary alcohols into carboxylic acids.
How do you convert primary alcohol to tertiary alcohol?
Answer: To produce a primary alcohol, the Grignard reagent is reacted with formaldehyde. Reacting a Grignard reagent with any other aldehyde will lead to a secondary alcohol. Finally, reacting a Grignard reagent with a ketone will generate a tertiary alcohol.
Does PCC affect double bond?
PCC oxidizes the alcohol (OH) group and does not affect any other functional group or double bond present in the compound [1-4].
What happens during oxidation of a tertiary alcohol quizlet?
Primary alcohols are oxidized to aldehydes, which undergo further oxidation to carboxylic acids. Secondary alcohols are oxidized to ketones, while tertiary alcohols do not undergo any oxidation at all.
Can tertiary alcohols be dehydrated?
Secondary and tertiary alcohols dehydrate through the E1 mechanism. Similarly to the reaction above, secondary and tertiary –OH protonate to form alkyloxonium ions. … Dehydration reaction of secondary alcohol: The dehydration mechanism for a tertiary alcohol is analogous to that shown above for a secondary alcohol.
Can a tertiary alcohol be reduced?
Benzylic alcohols, secondary alcohols, and tertiary alcohols were effectively reduced to give the corresponding alkanes in high yields. … This system showed high chemoselectivity only for the hydroxyl group while not reducing other functional groups that are readily reduced by standard reducing systems.
When tertiary alcohol reacts with KMnO4 What does it give?
Selina – Chemistry – Class 7 Tertiary alcohols on reaction with KMnO4 at elevated temperature form a mixture of carboxylic acids containing a lesser number of carbon atoms.
Does KMnO4 oxidize alcohol?
Potassium permanganate Under controlled conditions, KMnO4 oxidizes primary alcohols to carboxylic acids very efficiently. … KMnO4 will readily react with a carbon-carbon double bond before oxidizing a primary alcohol.
Can KMnO4 oxidize ketone?
Only very strong oxidizing agents such as potassium manganate(VII) (potassium permanganate) solution oxidize ketones. However, this type of powerful oxidation occurs with cleavage, breaking carbon-carbon bonds and forming two carboxylic acids. Because of this destructive nature this reaction is rarely used.
Does PCC oxidize alkenes?
As well as oxidizing (secondary alkyl)boranes to ketones,90,91 PCC oxidizes (primary alkyl)boranes to aldehydes in excellent yields (equation 47). The latter transformation cannot be accomplished with chromic acid. The reagent tolerates the presence of alkene, ester and acetal groups.
Is PCC a mild oxidizing agent?
Pyridinium chlorochromate (PCC) is a milder version of chromic acid. PCC oxidizes alcohols one rung up the oxidation ladder, from primary alcohols to aldehydes and from secondary alcohols to ketones. In contrast to chromic acid, PCC will not oxidize aldehydes to carboxylic acids. … Here are two examples of PCC in action.
What alcohol is difficult to oxidise?
It’s tough to oxidise tertiary alcohols. When tertiary alcohol is oxidised with powerful oxidising agents at high temperatures, the C-C bonds are broken, resulting in a mixture of carboxylic acids with fewer carbon atoms than the original 3° alcohol.
What does Na2Cr2O7 do in a reaction?
Sodium dichromate (Na2Cr2O7) or chromium trioxide (CrO3) will oxidize a primary alcohol to the carboxylic acid. Oxidation of primary alcohols to aldehydes can be accomplished by using pyridinium chlorochromate (PCC) as the oxidizing agent. PCC is a mixture is chromium trioxide, pyridine, and HCl.
What can h2cro4 oxidize?
One of the reagents that is commonly used for oxidation in organic chemistry is chromic acid. … Chromic acid, H2CrO4, is a strong acid and a reagent for oxidizing alcohols to ketones and carboxylic acids.
Can cyclohexanol be oxidized?
When cyclohexanol is exposed to sodium hypochlorite (NaOCl) and acetic acid, an oxidation reaction takes place that gives cyclohexanone as the product.
What reagent is used to oxidise alcohols?
The oxidising agent used in these reactions is normally a solution of sodium or potassium dichromate(VI) acidified with dilute sulphuric acid. If oxidation occurs, the orange solution containing the dichromate(VI) ions is reduced to a green solution containing chromium(III) ions.
Is neopentyl alcohol a tertiary alcohol?
As we know that carbinol is a primary alcohol which has a molecular formula of CH3−OH. The term ‘carbinol’ also known as methanol in its nomenclature. There’s a carbinol group in neopentyl alcohol and a tertiary group is also present. Hence, the derived name of neopentyl alcohol is Tertiary butyl alcohol.
Which of the following gives ketone on oxidation?
Secondary alcohols on oxidation give ketones.